Raptor Polar X: Separate a Wide Variety of Polar Analytes with a Novel Hybrid Stationary Phase
- No time-consuming derivatization or complex ion pairing.
- Switch between retention modes with simple mobile phase changes and short equilibration times.
- Ideal for increasing sensitivity and selectivity in LC-MS analyses.
Simplify the Analysis of Polar Compounds
Analyzing polar compounds using liquid chromatography has historically been a challenge. Poor retention and peak shape, complex mobile phases that may not be MS-friendly, long equilibration times, low sensitivity, and sample derivatization are all complications that reduce lab efficiency and productivity. However, the development of a novel column that is specifically designed for the analysis of a broad range of polar compounds allows scientists to avoid these problems by taking advantage of the true power of chromatography.
Providing the True Power of Chromatography
One of the most significant, yet least understood, aspects of method performance is the importance of using the proper stationary phase for a particular separation. For the analysis of polar compounds, reversed-phase columns do not provide adequate retention without the use of complex mobile phases or sample derivatization to compensate for the lack of effective interactions between the analytes and the column. But, by matching analytes to a stationary phase that provides purpose-built resolving power, you can avoid complex sample preparation procedures, save time and money, and reduce opportunities for error.
The Raptor Polar X stationary phase is specifically designed to selectively retain polar analytes through a balance of two retention mechanisms. This unique hybrid phase is ideal for analyzing a wide variety of polar compounds, especially when coupled with mass spectrometry. Simplify the analysis of polar compounds with the resolving power of Raptor Polar X columns from Restek.
Greater Than the Sum of Its Parts
Recognizing that the retention mechanisms most commonly used to retain polar compounds are hydrophilic interaction chromatography (HILIC) and ion-exchange approaches, Restek has developed a novel stationary phase chemistry that combines both modes on a single ligand. Because this unique ligand is bound to superficially porous particles, Raptor Polar X columns both reliably retain and efficiently separate a broad range of polar analytes.
Typical application-specific columns favor one retention mode over another, and gains made in the retention of one type of polar compound come at the expense of the performance for others. In contrast, with the Raptor Polar X column's patent-pending phase chemistry, two independent retention mechanisms are available for use, which provides truly balanced and flexible retention (Figure 1). Simple changes to mobile phase conditions allow analysts to switch between modes and selectively tune retention for the compounds of interest without needing long equilibration times before use or between samples.
Figure 1: The Raptor Polar X column can quickly and easily be switched between polar retention modes by simple changes in mobile phase conditions, providing an unprecedented ability to retain and separate a wide variety of polar compounds, even in the same analysis.
How It Works: Switching Retention Modes
When using a mobile phase with a relatively high-percentage of acetonitrile to water, a layer of water is formed at the surface of the silica into which polar compounds can partition. Partitioning into the water layer results in effective interactions between polar analytes and the ligand bound to the silica surface (Figure 2). The novel Raptor Polar X ligand chemistry makes this technique even easier, allowing for faster-than-ever column equilibration and re-equilibration. This means new columns are ready for use quickly, and sample throughput increases because less re-equilibration time is required between samples.
Figure 2: The rapid formation of a water layer at the silica surface allows a wide range of polar analytes to partition between the acetonitrile in the mobile phase and the water layer. This partitioning, along with interactions with the stationary phase, defines the HILIC retention mechanism.
For smaller polar compounds, these high-percentage acetonitrile conditions generate strong interactions between the analytes and the stationary phase, leading to excellent retention of small, highly polar, charged compounds. To fine-tune retention, simply increase the amount of water in the mobile phase. This will draw the charged, polar compounds into the mobile phase, effectively eluting them. When moving to a higher percent water mobile phase, the distinct HILIC partitioning retention will diminish and the stationary phase’s ion-exchange characteristics will become the dominant retention mechanism (Figure 3).
Figure 3: Under mobile phase conditions with higher water content, ion-exchange mechanisms take over as the dominant mode of retention for the analysis of polar compounds.
Retention Mode Switching: An Example
To illustrate these principles in action, Figure 4 demonstrates how two different types of polar compounds interact with the stationary phase, and how their responses are affected by simple mobile phase changes. In this example, we analyzed two water-soluble vitamins: vitamin B3 (niacin), which is an organic acid, and vitamin B1 (thiamine), which has a permanent positive charge.
In the top chromatogram, a highly organic mobile phase was used, which is favorable to HILIC separations. Using the Raptor Polar X column under these conditions, a water layer quickly forms on the surface of the silica particles. This allows polar compounds to partition from the acetonitrile layer into the water layer where they can also interact with the silica surface and the stationary phase. In this way, polar compounds are retained through hydrophilic interactions with the stationary phase and are still eluted under highly organic mobile phase conditions. This is very favorable for LC-MS/MS analysis because of the improved desolvation and increased ionization efficiency.
The bottom chromatogram highlights the chromatographic changes that are seen when the retention mechanism is switched by moving to a mobile phase with a higher percentage of water. As we move further away from ideal HILIC conditions, both vitamin B1 and vitamin B3 are less retained. This is because as the percentage of water in the mobile phase increases, conditions shift away from HILIC retention and toward the ion-exchange retention characteristics of the phase. The elution order also changes because vitamin B1 is less retained than vitamin B3 under ion-exchange conditions.
Figure 4: Simple changes to easy-to-prepare, MS-friendly mobile phases can emphasize or deemphasize the different retention mechanisms available with the novel Raptor Polar X phase chemistry.
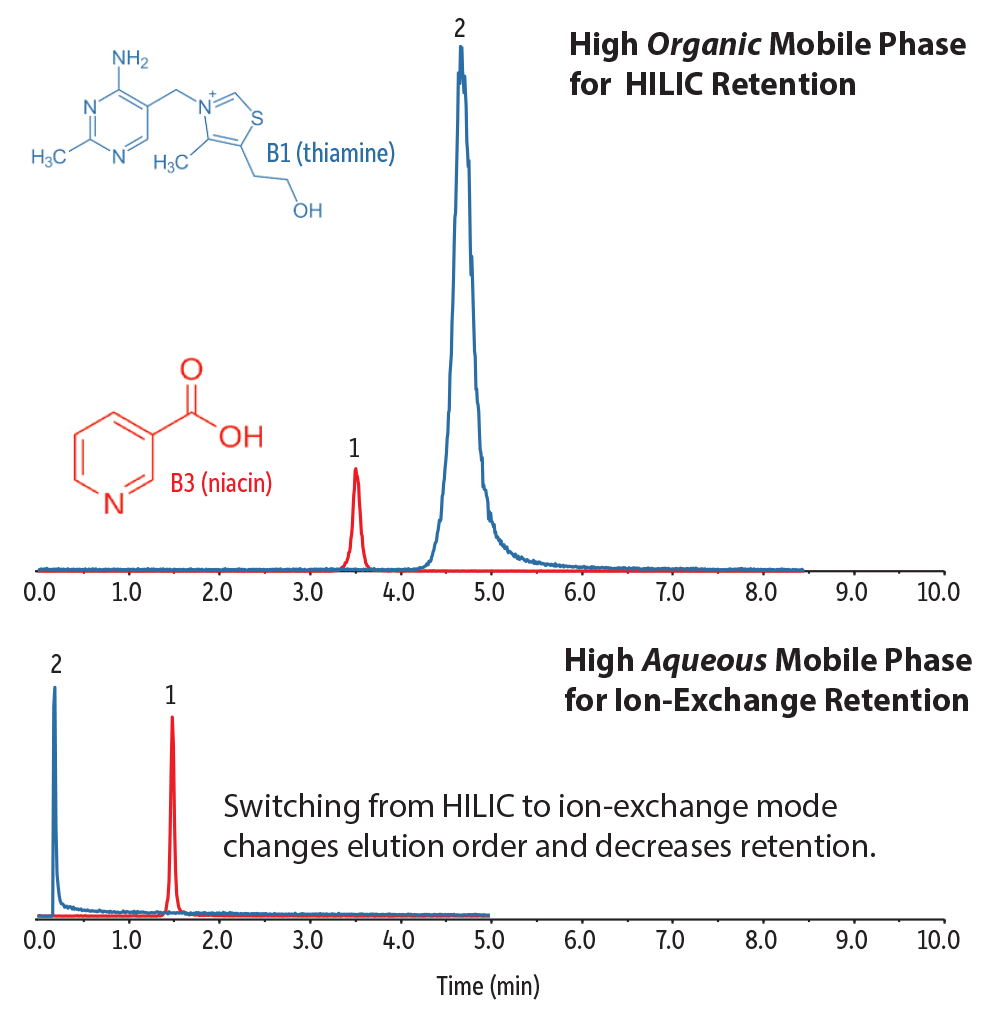
| Peaks | |
|---|---|
| 1. | Vitamin B3 |
| 2. | Vitamin B1 |
| Column | Raptor Polar X (cat.# 9311A52) |
|---|---|
| Dimensions: | 50 mm x 2.1 mm ID |
| Particle Size: | 2.7 µm |
| Temp.: | 30 °C |
| Standard/Sample | |
| Diluent: | 0.1% Formic acid in acetonitrile |
| Mobile Phase | |
| Flow: | 0.5 mL/min |
| Detector | MS/MS |
|---|---|
| Ion Source: | Electrospray |
| Ion Mode: | ESI+ |
| Instrument | HPLC |
| Notes | Mobile phase A: Water, 5 mM ammonium formate, 0.1% formic acid Mobile phase B: Acetonitrile, 0.1% formic acid Top chromatogram: 95% B, 10 min run, 2 μL injection (100 ppm B3, 0.01 ppm B1) Bottom chromatogram: 60% B, 5 min run, 0.5 μL injection (100 ppm B3, 0.1 ppm B1) |
Well-Balanced Hybrid Retention Lets You Analyze More Compounds with a Single Method
It is common for columns to specialize, to focus on a single type of interaction to the exclusion of others. This approach works well when the target analytes all share the same characteristics; however, polar compounds vary widely in their chemical traits. For the analysis of polar compounds, a specialized approach to column selection often means having to use multiple methods with different columns or conditions for each type of compound. As shown in the following examples, the hybrid phase chemistry of Raptor Polar X columns is a better alternative because the multimode retention profile allows a wide variety of target analytes to be analyzed in the same method.
QuPPe-Inspired List of Polar Contaminants
The European Quick Polar Pesticides (QuPPe) method includes a range of polar analytes from anionic polar herbicides, such as glyphosate and related compounds, to a variety of oxychloro contaminants, including chlorate and perchlorate. As shown in Figure 5, the Raptor Polar X column retains and quickly separates this diverse mix with the last compound eluting around 10.5 minutes and a total run time of only 13 minutes. This separation is performed with simple unbuffered mobile phases, acidified with 0.5% formic acid to reduce peak broadening and tailing. The method demonstrated in Figure 5 was also able to separate compounds that have similar mass fragments, like AMPA from N-acetyl AMPA and fosetyl aluminum from phosphonic acid and phosphoric acid (a commonly observed matrix component and source of interference).
Figure 5: In this analysis of polar compounds, a diverse group of analytes can all be successfully separated in a single run with the uniquely balanced retention characteristics of the Raptor Polar X column.
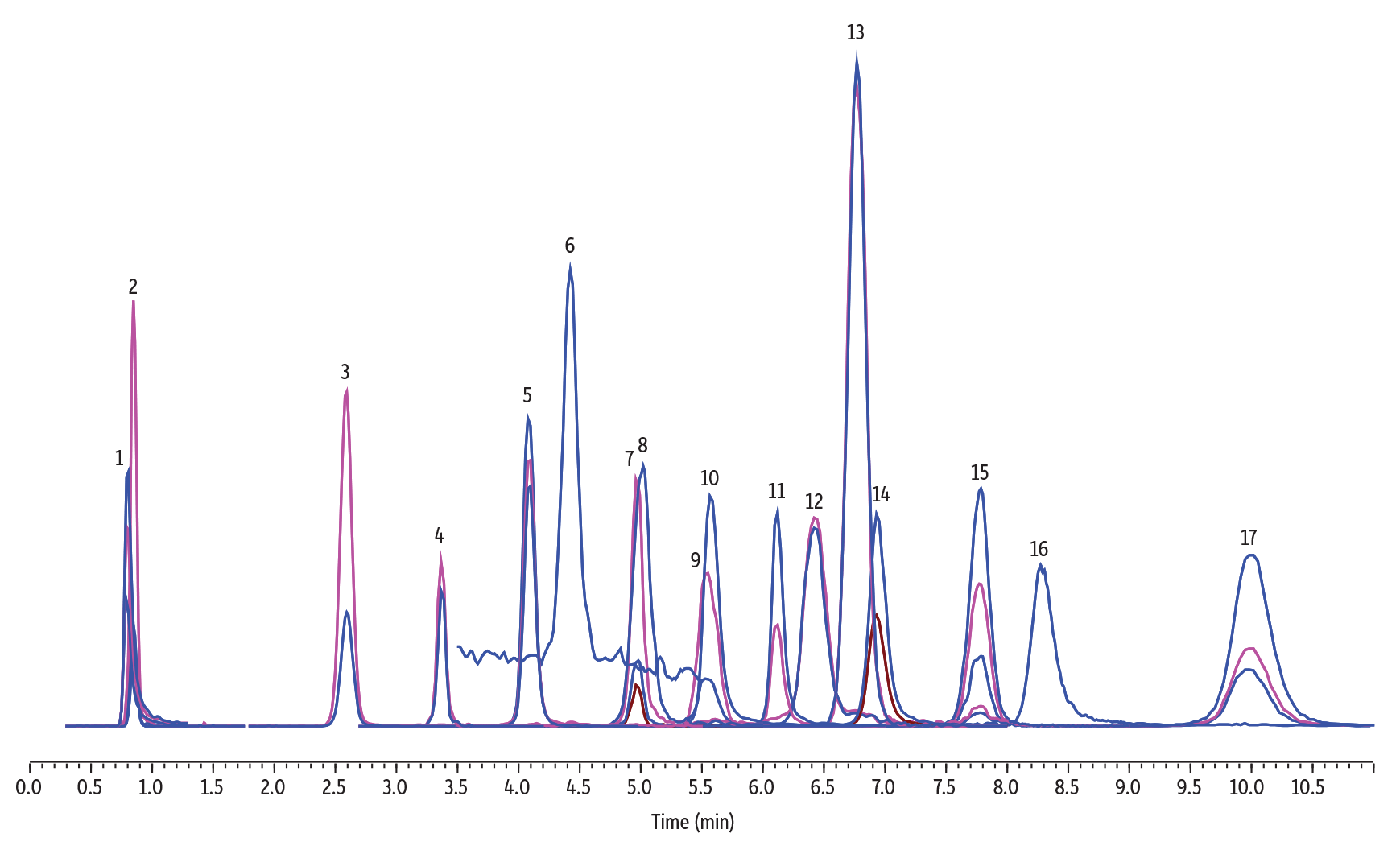
| Peaks | tR (min) | Conc. (ng/mL) | Precursor Ion | Product Ion 1 | Product Ion 2 | Product Ion 3 | Precursor 2 | Product Ion 2 | |
|---|---|---|---|---|---|---|---|---|---|
| 1. | Aminomethylphosphonic acid (AMPA) | 0.805 | 200 | 110.1 | 79.1 | 63.1 | 81.1 | - | - |
| 2. | Bialophos | 0.847 | 100 | 322.2 | 88.2 | 216.1 | 134.2 | - | - |
| 3. | Perchlorate | 2.593 | 5 | 101.0 | 84.95 | - | - | 98.9 | 83 |
| 4. | Glufosinate | 3.376 | 200 | 180.2 | 85.2 | 95.1 | - | - | - |
| 5. | 3-(Methylphophinico) propionic acid (MPPA) | 4.076 | 100 | 151.0 | 63.0 | 107.1 | 133.2 | - | - |
| 6. | Trifluoracetic acid (TFA) | 4.423 | 20 | 113.0 | 69.1 | 19.1 | - | - | - |
| 7. | 2-Hydroxyethane phosphonic acid (HEPA) | 4.969 | 100 | 125.1 | 79.0 | 95.0 | 63 | - | - |
| 8. | Difluoroacetic acid (DFA) | 5.018 | 200 | 95.0 | 51.1 | - | - | - | - |
| 9. | Chlorate | 5.542 | 100 | 85.0 | 69.0 | - | - | 83.0 | 67.1 |
| 10. | Ethephon | 5.564 | 200 | 143.1 | 107.2 | - | - | - | |
| 11. | Glyphosate | 6.113 | 200 | 168.1 | 63.1 | 79.1 | - | - | - |
| 12. | Bromide | 6.423 | 2000 | 80.9 | 80.9 | - | - | 79.0 | 79.0 |
| 13. | Bromate | 6.771 | 600 | 129.0 | 113 | - | - | 127 | 111.1 |
| 14. | N-acetyl AMPA | 6.932 | 200 | 152.1 | 110.1 | 62.9 | - | - | - |
| 15. | Fosetyl aluminum | 7.775 | 80 | 109.1 | 81.0 | 63.0 | 78.9 | - | - |
| 16. | Phosphonic acid | 8.275 | 500 | 81.1 | 62.9 | - | - | - | - |
| 17. | N-acetyl glufosinate | 9.980 | 200 | 222.2 | 136.1 | 134.1 | 59.0 | - | - |
| Column | Raptor Polar X (cat.# 9311A32) | ||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Dimensions: | 30 mm x 2.1 mm ID | ||||||||||||||||||||||||
| Particle Size: | 2.7 µm | ||||||||||||||||||||||||
| Pore Size: | 90 Å | ||||||||||||||||||||||||
| Temp.: | 35 °C | ||||||||||||||||||||||||
| Standard/Sample | |||||||||||||||||||||||||
| Diluent: | Water | ||||||||||||||||||||||||
| Inj. Vol.: | 1 µL | ||||||||||||||||||||||||
| Mobile Phase | |||||||||||||||||||||||||
| A: | Water, 0.5% formic acid | ||||||||||||||||||||||||
| B: | Acetonitrile, 0.5% formic acid | ||||||||||||||||||||||||
|
| Detector | MS/MS |
|---|---|
| Ion Mode: | ESI- |
| Mode: | MRM |
| Instrument | UHPLC |
In addition to the Restek-developed method shown in Figure 5, Raptor Polar X analytical and guard columns were independently evaluated and included in the QuPPe method itself [1]. They are used in a method for the detection of a wide variety of polar pesticide by LC-MS/MS in negative ESI mode.
Underivatized Amino Acids
Amino acids are a diverse group of very polar compounds that are commonly analyzed with reversed-phase or ion-exchange chromatography using pre- or post-column derivatization. Direct analysis of underivatized amino acids is challenging due to limited retention and poor chromatographic performance. However, underivatized amino acids with nonpolar, polar, positively charged, and negatively charged side chains are all retained and easily separated in one method on a Raptor Polar X column. Figure 6 shows the analysis of 21 amino acids, including a taurine supplement, in a liquid baby formula matrix using a simple protein precipitation and direct analysis of the resulting extract.
Figure 6: Compounds with various polarities, such as these amino acids, can all be analyzed using the same method on a Raptor Polar X column.
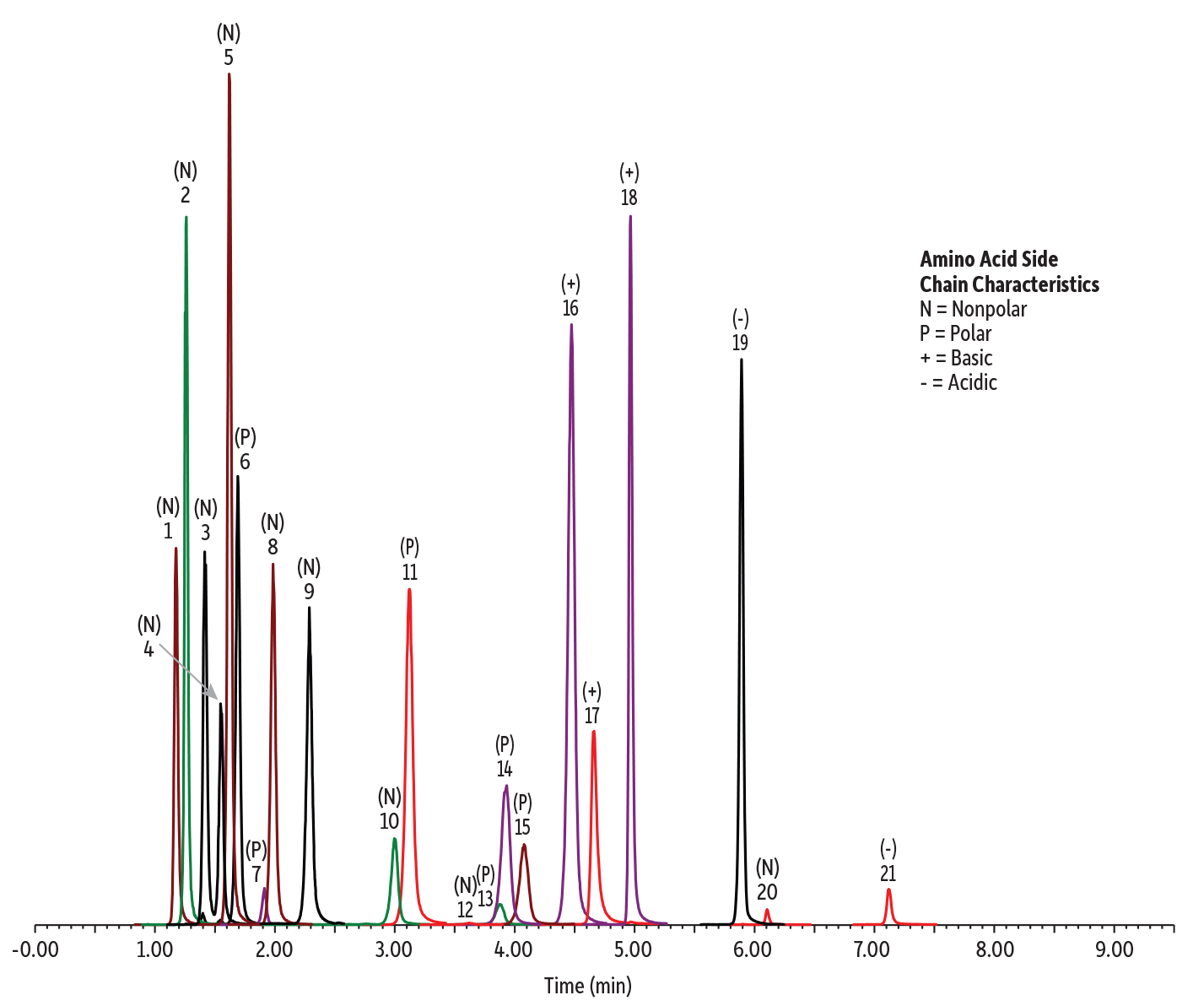
| Peaks | tR (min) | Precursor Ion | Product Ion | |
|---|---|---|---|---|
| 1. | Tryptophan | 1.17 | 205.07 | 146.08 |
| 2. | Phenylalanine | 1.26 | 166.13 | 120.10 |
| 3. | Leucine | 1.41 | 132.13 | 86.10 |
| 4. | Isoleucine | 1.55 | 132.13 | 86.10 |
| 5. | Methionine | 1.62 | 150.07 | 104.10 |
| 6. | Tyrosine | 1.69 | 182.10 | 136.08 |
| 7. | Taurine | 1.91 | 126.07 | 108.07 |
| 8. | Valine | 1.98 | 118.13 | 72.11 |
| 9. | Proline | 2.29 | 116.13 | 70.09 |
| 10. | Alanine | 3.00 | 90.03 | 44.10 |
| Peaks | tR (min) | Precursor Ion | Product Ion | |
|---|---|---|---|---|
| 11. | Threonine | 3.12 | 120.13 | 74.08 |
| 12. | Glycine | 3.62 | 76.10 | 30.11 |
| 13. | Glutamine | 3.87 | 147.13 | 84.07 |
| 14. | Serine | 3.93 | 106.07 | 60.09 |
| 15. | Asparagine | 4.08 | 133.13 | 74.07 |
| 16. | Arginine | 4.47 | 175.17 | 70.09 |
| 17. | Histidine | 4.66 | 156.07 | 110.16 |
| 18. | Lysine | 4.97 | 147.13 | 84.13 |
| 19. | Glutamic acid | 5.89 | 148.10 | 84.10 |
| 20. | Cystine | 6.10 | 241.13 | 152.00 |
| 21. | Aspartic acid | 7.12 | 134.07 | 74.06 |
| Column | Raptor Polar X (cat.# 9311A12) | ||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Dimensions: | 100 mm x 2.1 mm ID | ||||||||||||||||||||||||
| Particle Size: | 2.7 µm | ||||||||||||||||||||||||
| Temp.: | 30 °C | ||||||||||||||||||||||||
| Standard/Sample | |||||||||||||||||||||||||
| Diluent: | 20:80 Water:acetonitrile, 0.01 N HCl | ||||||||||||||||||||||||
| Conc.: | Endogenous amino acids | ||||||||||||||||||||||||
| Inj. Vol.: | 5 µL | ||||||||||||||||||||||||
| Mobile Phase | |||||||||||||||||||||||||
| A: | Water, 0.5% formic acid | ||||||||||||||||||||||||
| B: | Mobile Phase B: 9:1 Acetonitrile:20 mM ammonium formate in water (pH 3.0) (The ammonium formate concentration is 20 mM relative to the total volume of mobile phase B. See preparation notes for instructions on diluting a 200 mM aqueous starting solution.) | ||||||||||||||||||||||||
|
| Detector | MS/MS |
|---|---|
| Ion Mode: | ESI+ |
| Mode: | MRM |
| Instrument | UHPLC |
| Sample Preparation | A 200 µL aliquot of protein hydrolysate formula (Similac ALIMENTUM) was mixed with 800 µL of acetonitrile and 10 µL of 1 N HCl. After centrifugation at 4000 rpm for 5 minutes, the supernatant was diluted 20-fold with 20:80 water:acetonitrile (0.01 N HCl) and injected for analysis. |
| Notes | Mobile Phase B Preparation: To make 500 mL of mobile phase B, measure ~45 mL of water into a small beaker and add 1 mL of 10 M ammonium formate solution. Adjust pH to 3.0 by adding formic acid and then bring the volume to 50 mL with water. Combine this 50 mL ammonium formate solution (pH 3.0) with 450 mL of acetonitrile to complete the preparation. |
Ultrashort- to Long-Chain PFAS
The last illustration of the Raptor Polar X column’s ability to maximize method productivity is an application that paves a path for future testing. Current LC-MS/MS methods for per- and polyfluoroalkyl substances (PFAS) analysis are focused on short-chain (C4-C6), long-chain (C8 and up), and alternative compounds and do not include emerging ultrashort-chain (C2 and C3) compounds. Typical reversed-phase methods for PFAS analysis do not have sufficient retention for ultrashort-chain PFAS; whereas other methods based on anionic exchange often have too much retention, resulting in poor chromatographic performance. Because of the balanced retention mechanisms found in the Raptor Polar X column, ultrashort-chain PFAS and long chain PFAS can all be analyzed in a single 5-minute isocratic run (Figure 7).
Figure 7: Ultrashort-chain, legacy, and alternative PFAS analyzed with a single method on the novel Raptor Polar X stationary phase.
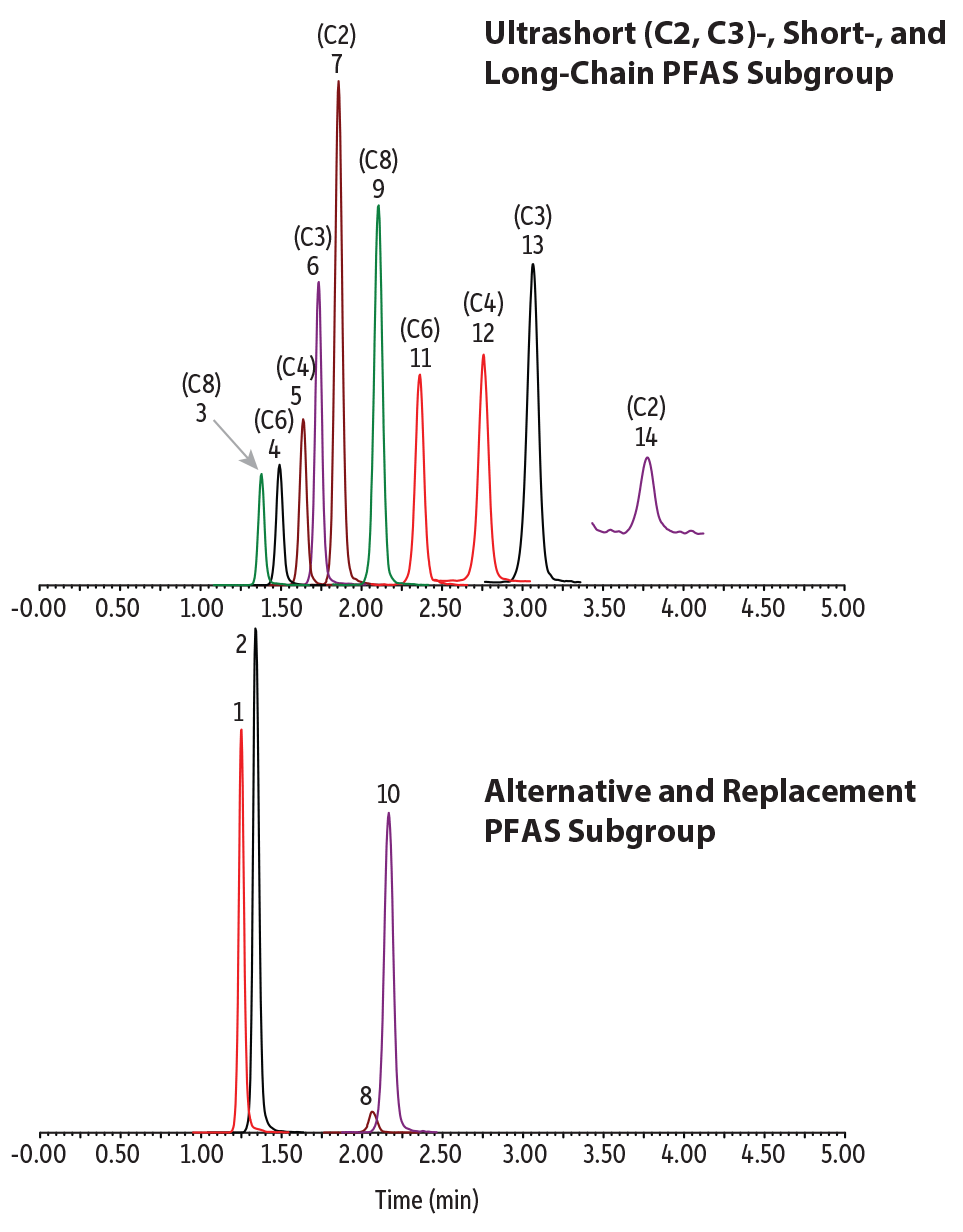
| Peaks | tR (min) | Conc. (ng/L) | Precursor Ion | Product Ion | |
|---|---|---|---|---|---|
| 1. | 11-Chloroeicosafluoro-3-oxanonane-1-sulfonate (11CL-PF3OUdS) | 1.25 | 400 | 630.78 | 450.80 |
| 2. | 9-Chlorohexadecafluoro-3-oxanonane-1-sulfonate (9Cl-PF3ONS) | 1.34 | 400 | 530.78 | 350.85 |
| 3. | Perfluorooctanesulfonic acid (PFOS) | 1.38 | 400 | 498.84 | 79.97 |
| 4. | Perfluorohexanesulfonic acid (PFHxS) | 1.49 | 400 | 398.90 | 79.97 |
| 5. | Perfluorobutanesulfonic acid (PFBS) | 1.64 | 400 | 298.97 | 79.97 |
| 6. | Perfluoropropanesulfonic acid (PFPrS) | 1.73 | 400 | 248.97 | 79.98 |
| 7. | Perfluoroethanesulfonic acid (PFEtS) | 1.86 | 400 | 198.98 | 79.92 |
| Peaks | tR (min) | Conc. (ng/L) | Precursor Ion | Product Ion | |
|---|---|---|---|---|---|
| 8. | Hexafluoropropylene oxide dimer acid (HFPO-DA) | 2.06 | 400 | 284.97 | 168.92 |
| 9. | Perfluorooctanoic acid (PFOA) | 2.11 | 400 | 412.90 | 368.91 |
| 10. | Ammonium 4,8-dioxa-3H-perfluorononanoate (ADONA) | 2.15 | 400 | 376.90 | 250.93 |
| 11. | Perfluorohexanoic acid (PFHxA) | 2.36 | 400 | 312.97 | 268.90 |
| 12. | Perfluorobutanoic acid (PFBA) | 2.76 | 400 | 212.97 | 168.97 |
| 13. | Perfluoropropionic acid (PFPrA) | 3.06 | 400 | 163.03 | 119.01 |
| 14. | Trifluoroacetic acid (TFA) | 3.77 | 400 | 113.03 | 69.01 |
| Column | Raptor Polar X (cat.# 9311A52) | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Dimensions: | 50 mm x 2.1 mm ID | ||||||||||||
| Particle Size: | 2.7 µm | ||||||||||||
| Temp.: | 40 °C | ||||||||||||
| Standard/Sample | |||||||||||||
| Diluent: | 50:50 Water:methanol | ||||||||||||
| Conc.: | 400 ng/L | ||||||||||||
| Inj. Vol.: | 10 µL | ||||||||||||
| Mobile Phase | |||||||||||||
| A: | Water, 10 mM ammonium formate, 0.05% formic acid | ||||||||||||
| B: | 60:40 Acetonitrile:methanol, 0.05% formic acid | ||||||||||||
|
| Detector | MS/MS |
|---|---|
| Ion Mode: | ESI- |
| Mode: | MRM |
| Instrument | UHPLC |
To Passivate or Not to Passivate?
Glyphosate can be difficult to analyze at trace levels because it chelates with active metal sites in an LC system. Even though the Raptor Polar X column contains a treated sample flow path and arrives in your lab ready for initial use, metal components along the sample flow path through the LC instrument may benefit from passivation. Whether or not passivation is required will depend on the application and on your particular instrument setup, but for the analysis of polar compounds that are known to chelate, such as glyphosate, Restek recommends treating your system with LC passivation solution (cat.# 32475) prior to running samples.
A Novel Stationary Phase Built on the Time-Tested Foundation of Raptor Quality
The Raptor Polar X has been designed, manufactured, and tested to the same exacting standards that have made Raptor columns synonymous with quality. Whether it’s robust injection-to-injection performance when analyzing real-world samples in challenging matrices like spinach (Figure 8) or reliable column-to-column performance when it is eventually time to replace the column (Figure 9), you can count on Raptor brand quality. In addition, the single ligand approach to the Raptor Polar X column’s hybrid retention functionality helps ensure that each column is made uniformly and can withstand even aggressive conditions such as large volume water injections without loss of stationary phase.
Figure 8: Excellent peak shape and retention time stability after hundreds of injections of a spinach extract demonstrate the robust performance of Raptor Polar X columns for the analysis of polar compounds.
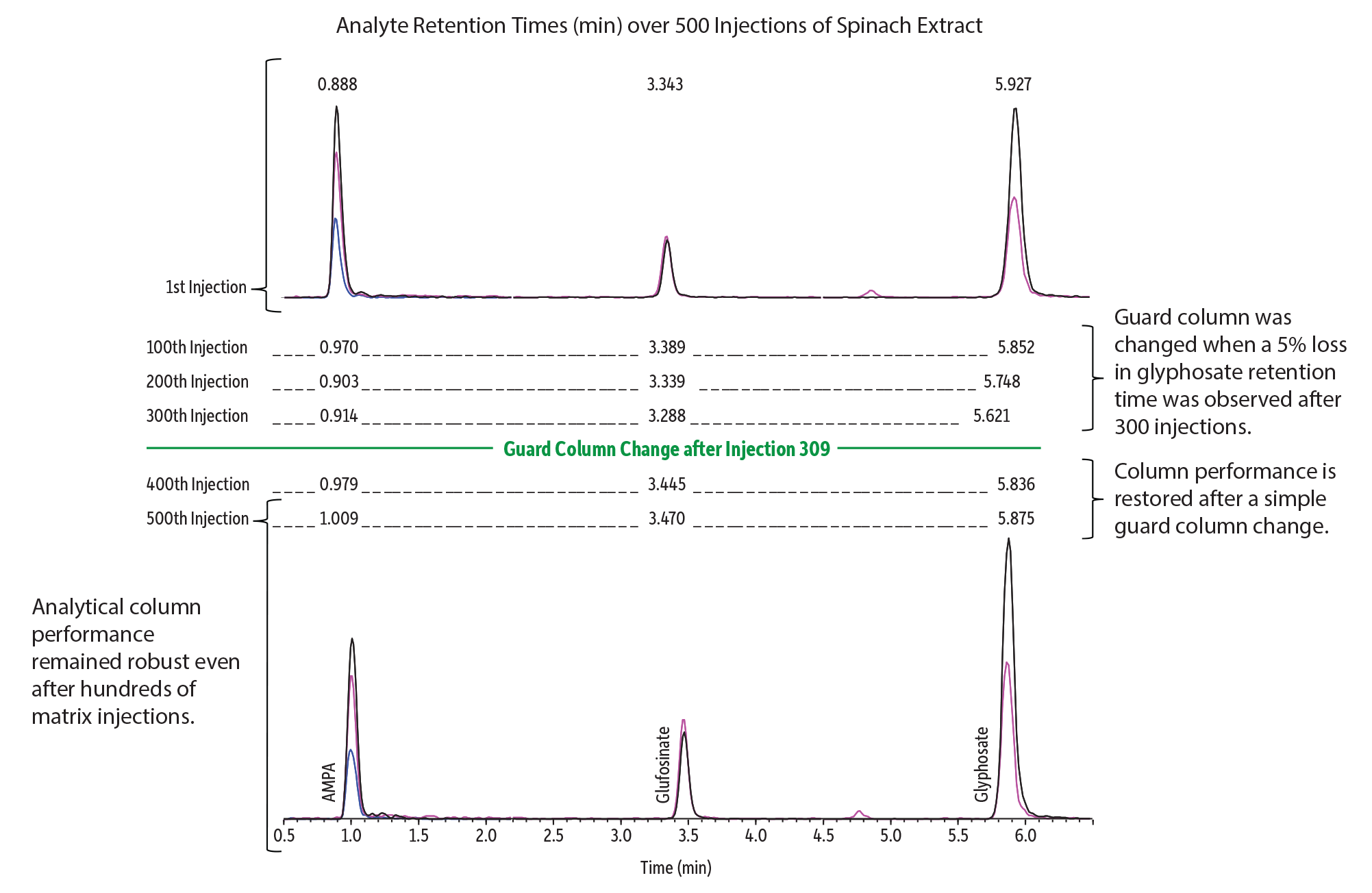
| Peaks | Conc. (ng/mL) | Precursor Ion | Product Ion 1 | Product Ion 2 | Product Ion 3 | |
|---|---|---|---|---|---|---|
| 1. | AMPA | 100 | 110.10 | 79.05 | 63.10 | 81.10 |
| 2. | Glufosinate | 100 | 180.20 | 85.15 | 95.10 | - |
| 3. | Glyphosate | 100 | 168.10 | 63.05 | 79.05 | - |
| Column | Raptor Polar X (cat.# 9311A32) | ||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Dimensions: | 30 mm x 2.1 mm ID | ||||||||||||||||||||||||
| Particle Size: | 2.7 µm | ||||||||||||||||||||||||
| Pore Size: | 90 Å | ||||||||||||||||||||||||
| Guard Column: | Raptor Polar X guard column cartridge 5 mm, 2.1 mm ID, 2.7 µm (cat.# 9311A0252) | ||||||||||||||||||||||||
| Temp.: | 35 °C | ||||||||||||||||||||||||
| Standard/Sample | Spinach extract | ||||||||||||||||||||||||
| Inj. Vol.: | 2 µL | ||||||||||||||||||||||||
| Mobile Phase | |||||||||||||||||||||||||
| A: | Water, 0.5% formic acid | ||||||||||||||||||||||||
| B: | Acetonitrile, 0.5% formic acid | ||||||||||||||||||||||||
|
| Detector | MS/MS |
|---|---|
| Ion Mode: | ESI- |
| Mode: | MRM |
| Instrument | UHPLC |
| Sample Preparation | Frozen spinach was added to a Blixer processor with dry ice (3:1-4:1 ratio) and then ground into a very fine powder. The homogenate was placed into the freezer immediately. A 5.0 gram sample of the spinach powder was weighed into a 50 mL centrifuge tube (cat.# 25846). According to the QuPPe method (Quick Polar Pesticides Method), 5.0 mL of methanol with 1.0% formic acid was added into the centrifuge tube. The tube was shaken by hand for 1 min and then by a mechanical shaker vigorously for 5 min. After centrifuging for 10 min at 4200 rpm, the supernatant was filtered through a 0.22 μm filter (cat.# 23984). The final extract was fortified with the AMPA, glufosinate, and glyphosate at a final concentration of 100 ng/mL. |
Figure 9: Raptor Polar X columns provide reliable lot-to-lot reproducibility.
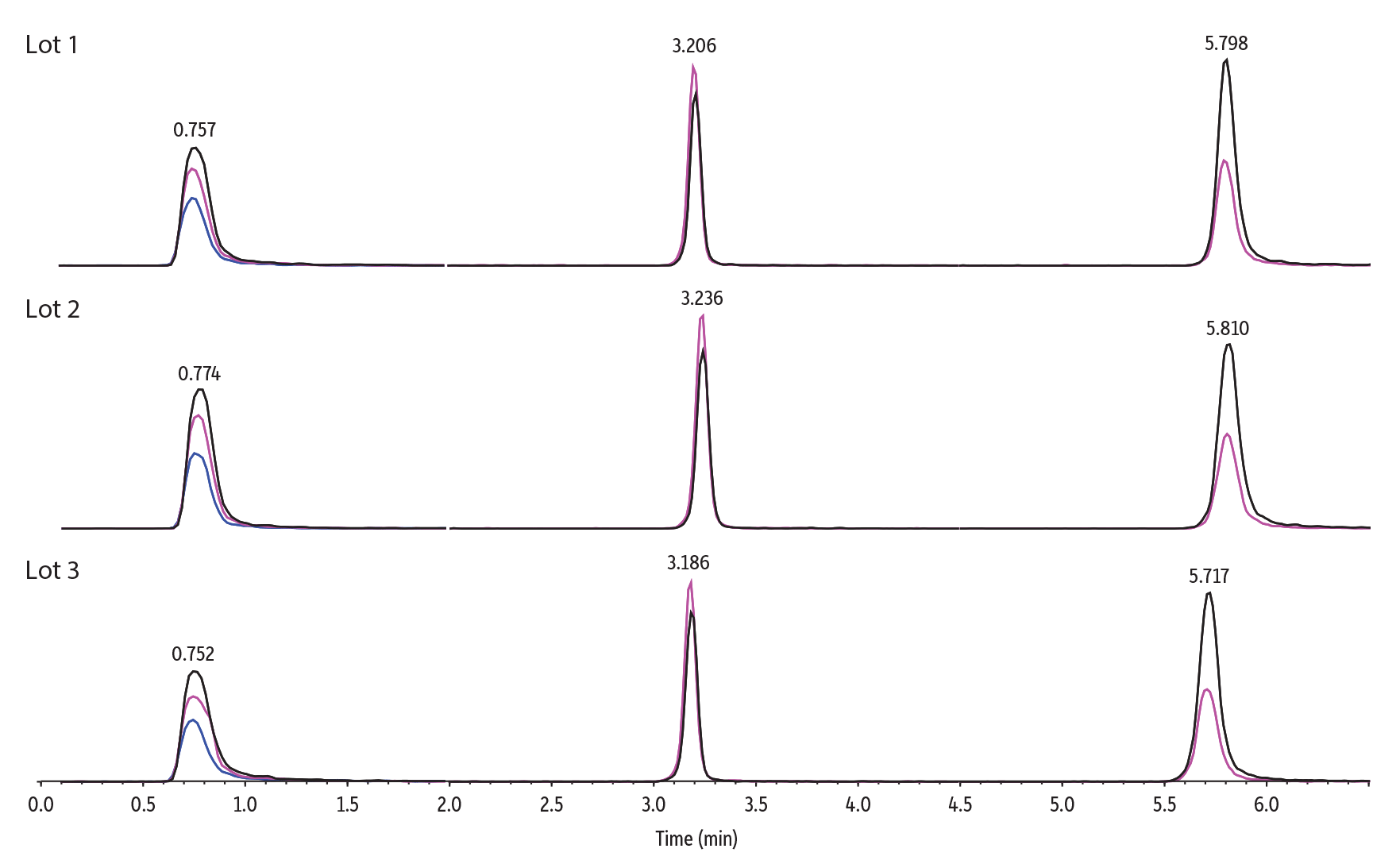
| Peaks | Conc. (ng/mL) | Precursor Ion | Product Ion 1 | Product Ion 2 | Product Ion 3 | |
|---|---|---|---|---|---|---|
| 1. | AMPA | 100 | 110.10 | 79.05 | 63.10 | 81.10 |
| 2. | Glufosinate | 100 | 180.20 | 85.15 | 95.10 | - |
| 3. | Glyphosate | 100 | 168.10 | 63.05 | 79.05 | - |
| Column | Raptor Polar X (cat.# 9311A32) | ||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Dimensions: | 30 mm x 2.1 mm ID | ||||||||||||||||||||||||
| Particle Size: | 2.7 µm | ||||||||||||||||||||||||
| Pore Size: | 90 Å | ||||||||||||||||||||||||
| Temp.: | 35 °C | ||||||||||||||||||||||||
| Standard/Sample | |||||||||||||||||||||||||
| Diluent: | Water | ||||||||||||||||||||||||
| Inj. Vol.: | 5 µL | ||||||||||||||||||||||||
| Mobile Phase | |||||||||||||||||||||||||
| A: | Water, 0.5% formic acid | ||||||||||||||||||||||||
| B: | Acetonitrile, 0.5% formic acid | ||||||||||||||||||||||||
|
| Detector | MS/MS |
|---|---|
| Ion Mode: | ESI- |
| Mode: | MRM |
| Instrument | UHPLC |
Raptor Polar X Columns: Revolutionizing the LC Analysis of Polar Compounds
Poor retention, low response, complex sample preparation procedures…the analysis of polar compounds historically has posed challenges to both data quality and lab productivity. Now, Raptor Polar X columns—with their novel phase chemistry that blends HILIC and ion-exchange retention on a single ligand—significantly simplify the analysis of these difficult and varied compounds. And, by bonding that unique ligand to superficially porous particles (SPP), Restek has created a phase that retains and efficiently separates a wide variety of polar compounds. The synergy created by these features results in an unparalleled ability to analyze a wide variety of polar compounds, providing a powerful, productive, and versatile tool for LC scientists across many industries.
References
- M. Anastassiades, A.-K. Wachtler, D. I. Kolberg, E. Eichhorn, H. Marks, A. Benkenstein, S. Zechmann; D. Mack, C. Wildgrube, A. Barth, I. Sigalov, S. Görlich, D. Dörk, G. Cerchia, Quick method for the analysis of highly polar pesticides in food involving extraction with acidified methanol and LC - or ICMS/MS Measurement - I. Food of Plant Origin (QuPPe-PO-Method)–Version 12 (published on EURL-SRM website on July 23, 2021). https://www.eurl-pesticides.eu/docs/public/tmplt_article.asp?LabID=200&CntID=1115&Theme_ID=1&Pdf=False&Lang=EN

