GC-MS Analysis of Phthalates: Comparison of GC Stationary Phase Performance
Abstract
Phthalates are ubiquitous in the environment and have attracted attention due to their potential adverse impact on human health. For this reason, detection and separation of phthalates has become a necessity. Gas chromatography is an effective approach for separating phthalates, and it can be paired with several detection techniques, including electron capture (ECD), flame ionization (FID), and mass spectrometry (MS). In this study, Pro EZGC gas chromatographic modeling software was employed to determine the optimal stationary phases and conditions for phthalate GC-MS analysis. The separation of phthalates was compared on seven different stationary phases: Rtx-440, Rxi-XLB, Rxi-5ms, Rtx-50, Rxi-35Sil MS, Rtx-CLPesticides, and Rtx-CLPesticides2 phases. In all cases, 18 EPA- and EU-listed phthalates were analyzed in less than 6 minutes. In addition, an extended list of 37 phthalates was analyzed in less than 40 minutes using an optimized method. Both Rtx-440, which is unique to Restek, and Rxi-XLB columns showed the best resolution of the complex phthalate mixture.
Introduction
Phthalates are widely used as plasticizers in a variety of industry products. However, some phthalates are considered to be endocrine disruptors [1] and are associated with a number of problems, including birth defects [2]; high blood pressure in children [3]; pregnancy-induced hypertensive heart diseases [4]; respiratory problems [5]; and obesity [6]. The European Union (EU) and United States Environmental Protection Agency (U.S. EPA) have restricted the use of the most harmful phthalates (Table I).
GC-MS is a commonly used technique for phthalate analysis because it is simple, fast, and inexpensive. GC-MS also provides mass spectral information, making it a powerful instrumental platform for phthalate determination. Using a GC column that provides good chromatographic separations is important because structural similarities among phthalates can make MS identification and quantification challenging. For instance, many phthalates share a common base peak ion (m/z 149), which makes identification and quantification of coeluting phthalates difficult. Technical grade mixtures and isomers further complicate the problem.
A recently published review summarized the most used GC and LC columns for phthalate analysis [7]. According to the literature, GC-MS has better chromatographic resolution compared to LC-MS for phthalate determination. The most commonly employed GC columns in descending order of popularity are 5-type, XLB-type, 35-type, 17-type, 50-type, and 1-type. The separation achieved on any stationary phase can be refined by adjusting instrument conditions, but this can be a time-consuming process when done in the laboratory. Pro EZGC modeling software can quickly optimize GC parameters (e.g., carrier gas type, flow rate, temperature program, column dimensions, and guard column) to produce the shortest analysis time on a given stationary phase. In this study, libraries of 37 phthalates (Table II) were built into the Pro EZGC program for seven stationary phases: Rtx-440, Rxi-XLB, Rxi-5ms, Rtx-50, Rxi-35Sil MS, Rtx-CLPesticides, and Rtx-CLPesticides2 phases. These stationary phases were evaluated for the analysis of both regulated and unregulated phthalates.
Experimental Chemicals
Restek’s EPA Method 8061A phthalates esters mixture (cat.# 33227), which contains 15 of the target analytes, each at a concentration of 1,000 μg/mL, was used as a primary reference standard. Benzyl benzoate (cat.# 31847) was used as the internal standard. All other phthalate standards were purchased from Chem Service.
Instrument
GC-MS analysis was performed on a Shimadzu QP2010 Plus GC-MS. The instrument was equipped with one of seven Restek columns using 30 m × 0.25 mm × 0.25 µm dimensions (0.20 µm for the Rtx-CLPesticides2 column). Windows-based Pro EZGC ROM software was used to determine optimized conditions for each column and then all columns were directly compared by analyzing samples under the single set of conditions that had produced the best overall separation. This direct comparison provides a better understanding of the differences in selectivity among the columns. Detailed column descriptions and GC-MS experimental parameters are given in Table I and Table III, respectively.
Sample Preparation
Standards were dissolved and diluted in methylene chloride. Standard solutions were prepared at 50 μg/mL (80 μg/mL for the internal standard benzyl benzoate). During sample preparation, plastics were strictly avoided; all preparation work was performed using glassware (volumetric flasks, syringes, vials, etc.).
Results and Discussion
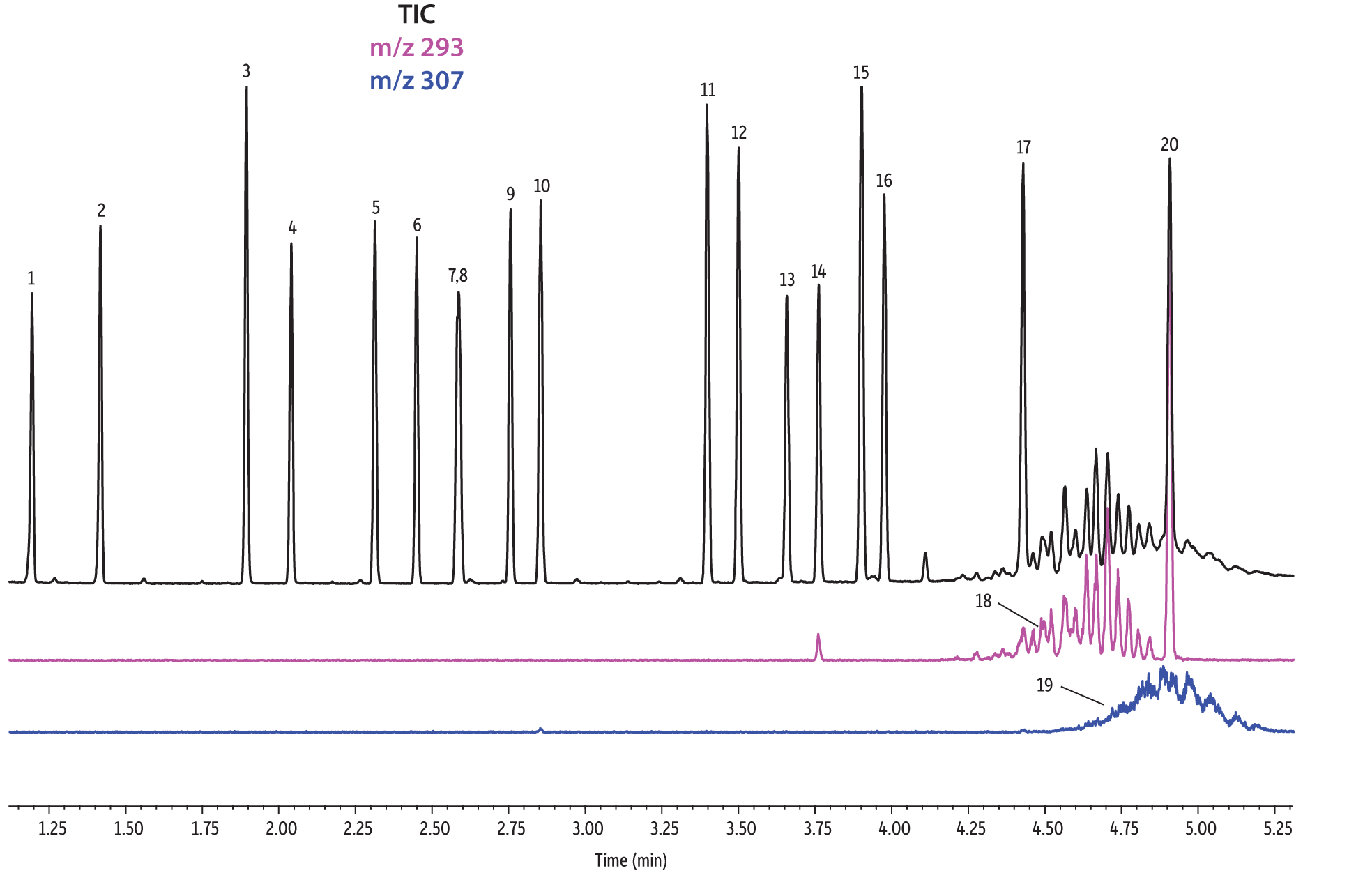
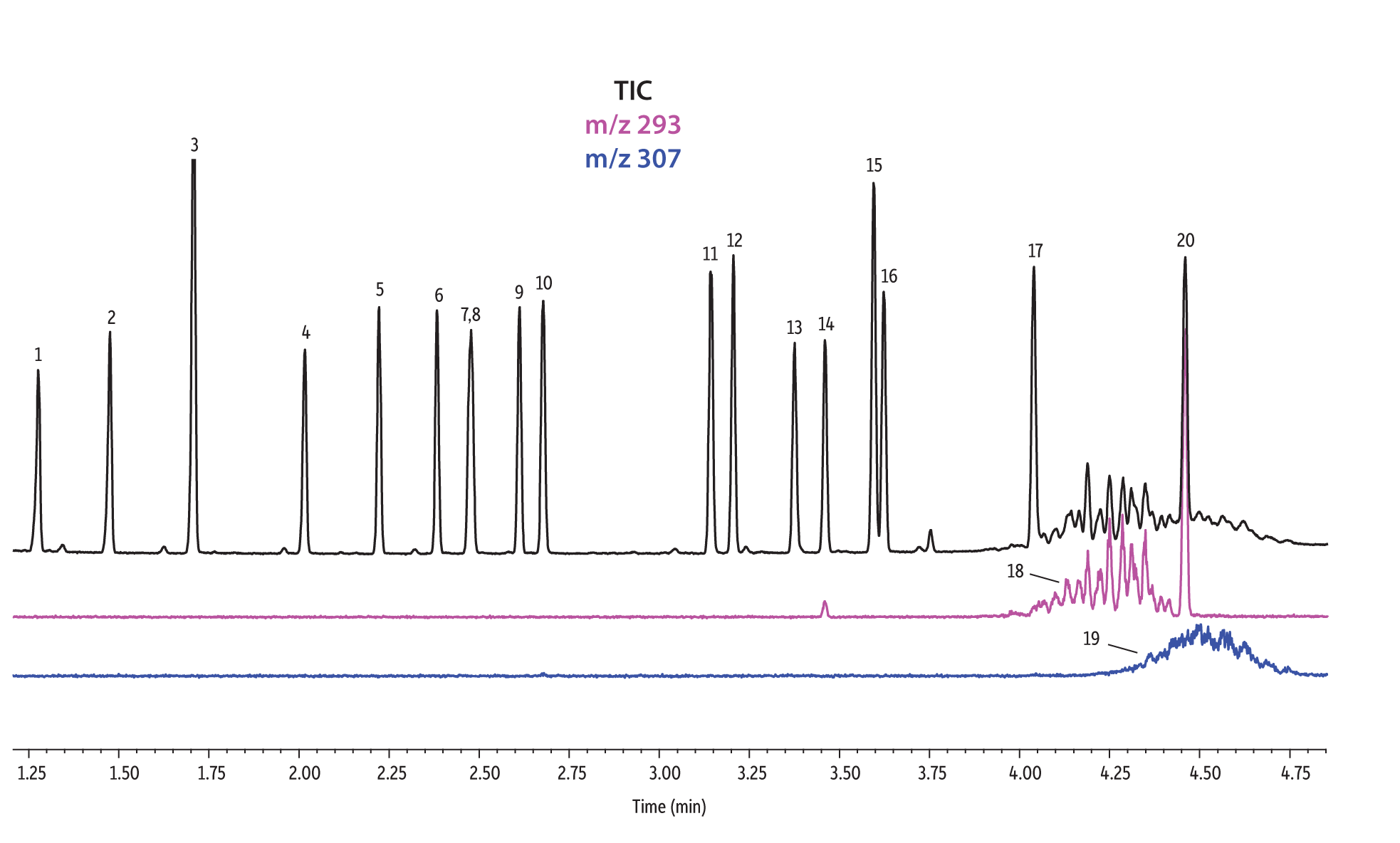
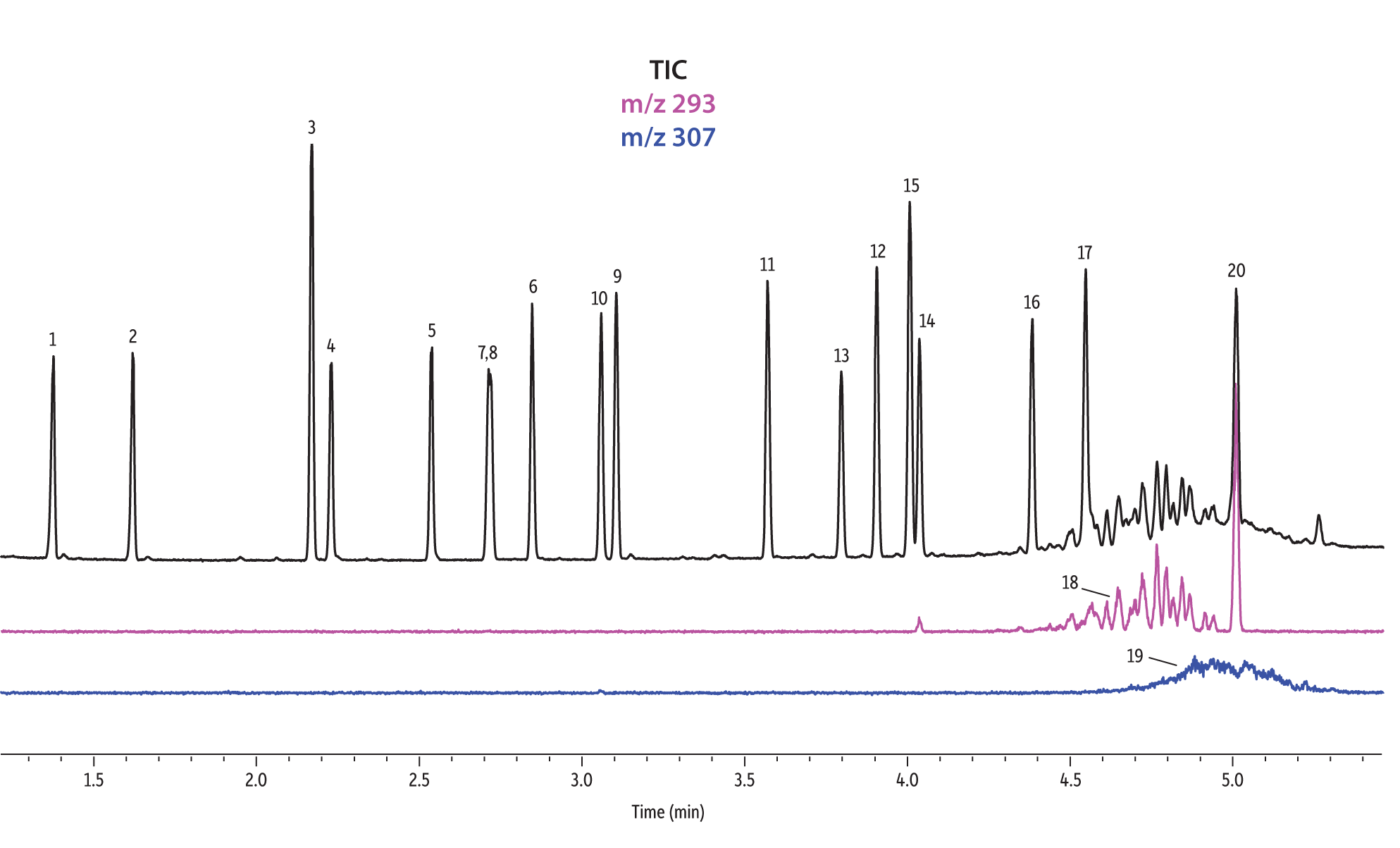
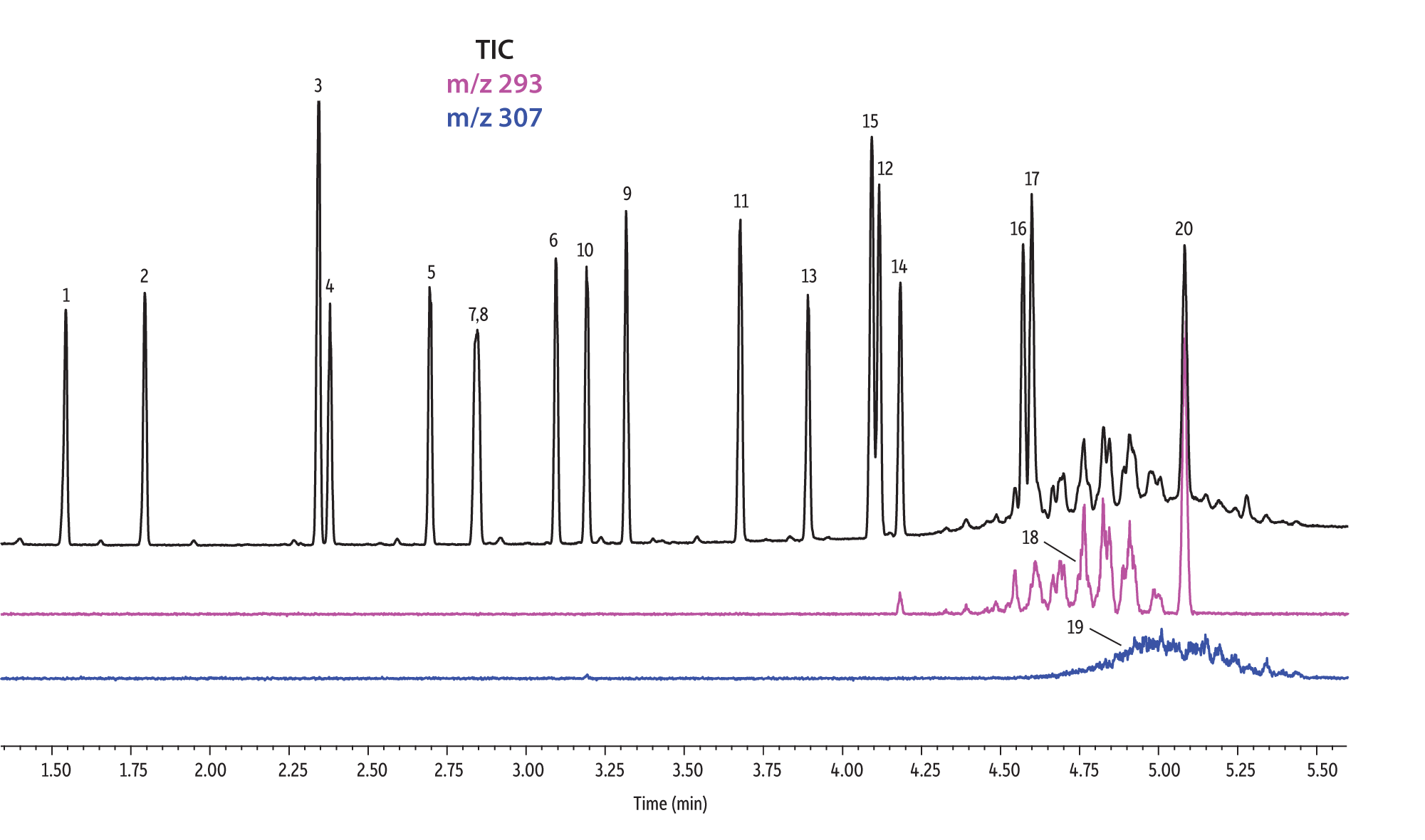
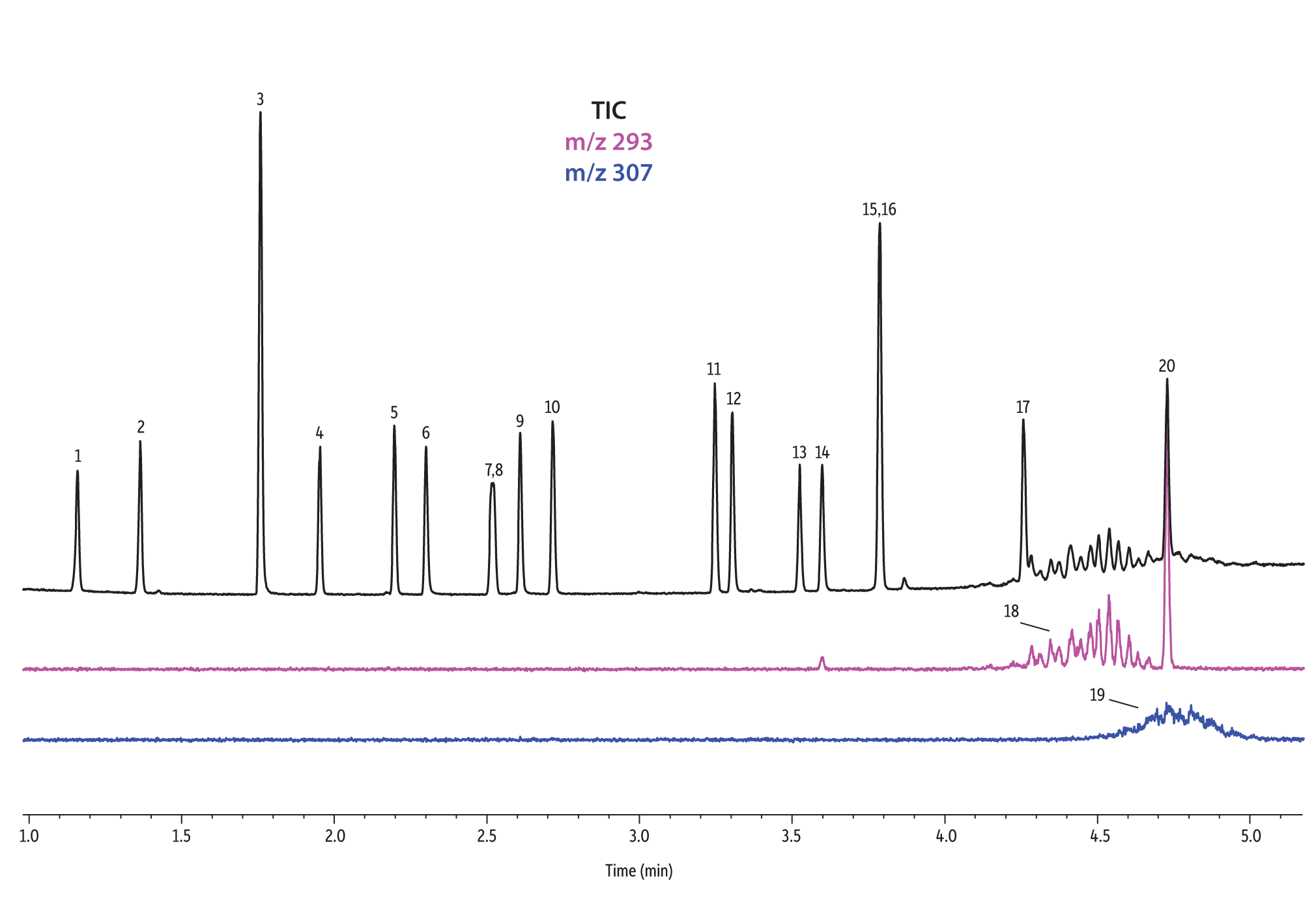
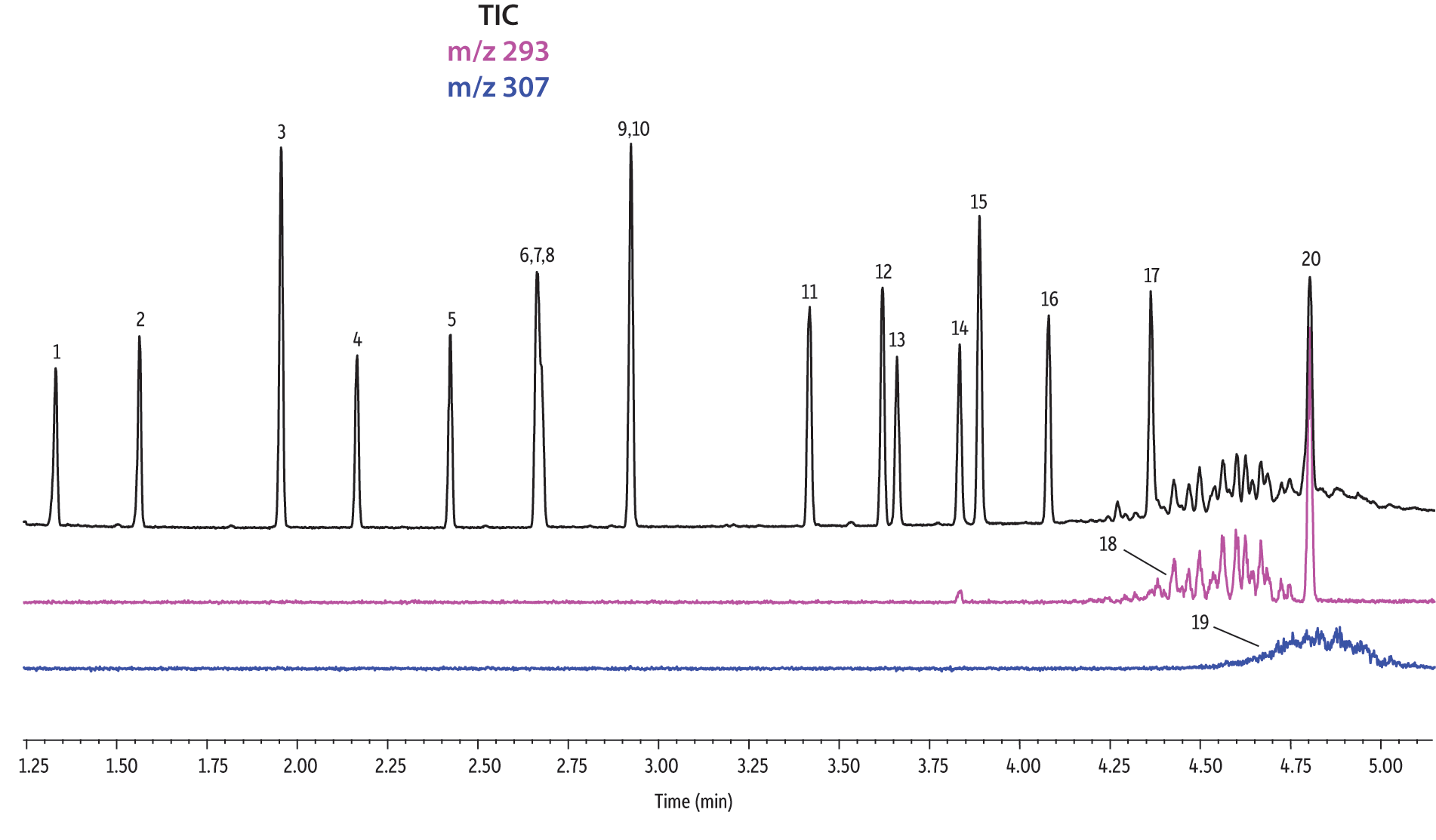
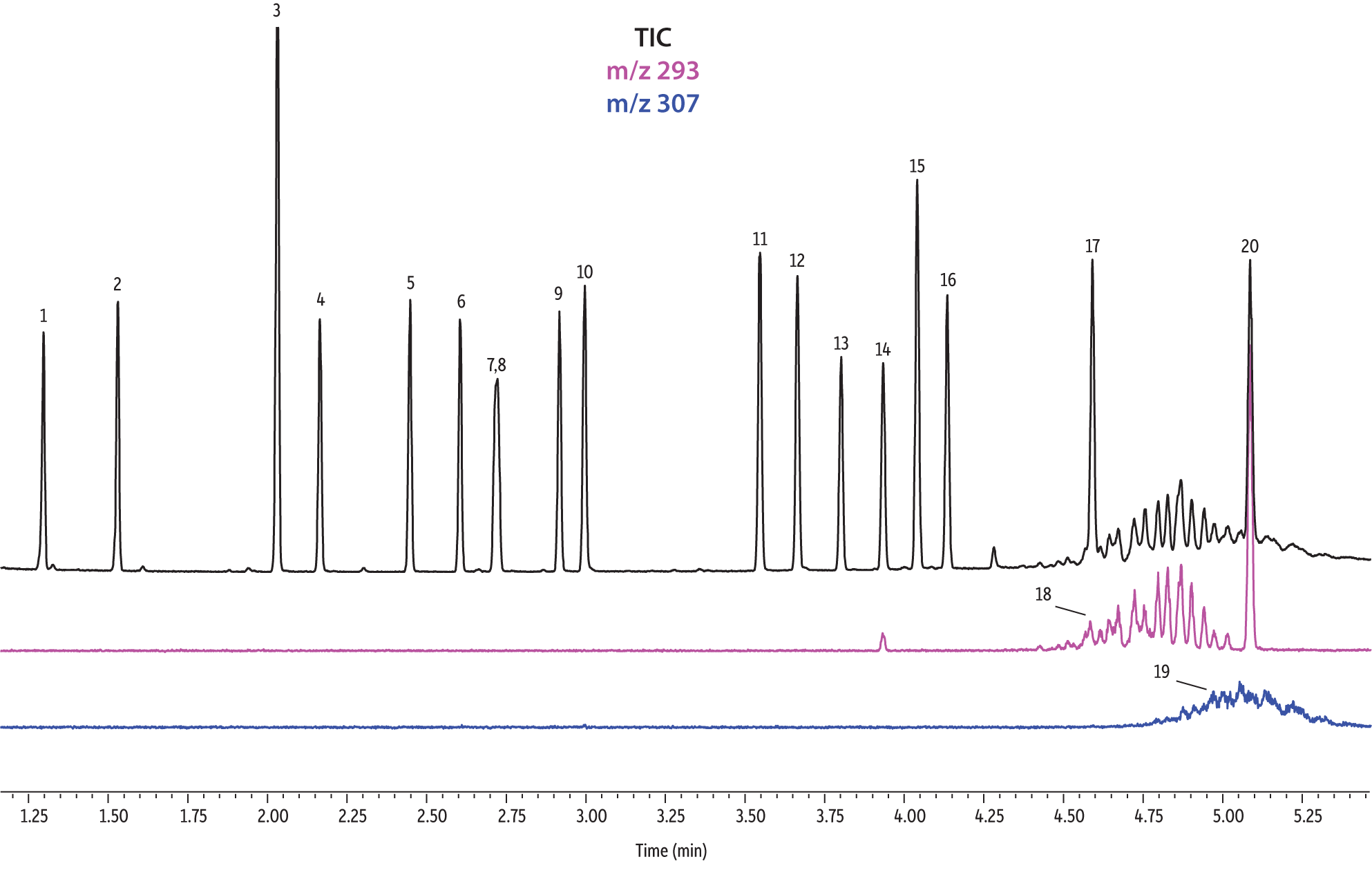
A direct comparison of column performance for the separation of EPA- and EU-regulated phthalates was performed. Phthalate retention times on seven different phases were predicted by the Pro EZGC ROM program under the same GC conditions. These conditions, shown in Table I, were the conditions that produced the best overall chromatographic results among the separate sets of conditions that had been optimized for each column. Coelutions were defined as compound pairs with a resolution value of less than 1.5. The total analysis time was less than 6 minutes. In order to confirm the retention time predictions made by the Pro EZGC ROM software, chromatograms were collected on each stationary phase under the same conditions used by the software (Figure 1). Because the column lengths were not exactly 30 meters long, as they were in the simulation, the absolute retention times were slightly different from the predicted values; however, the elution orders and coeluting pairs were exactly the same as predicted. Rtx-440, Rxi-XLB, Rtx-CLPesticides, and Rxi-35Sil MS columns provided baseline separation for all EPA- and EU-listed phthalates. The two isomers of bis[4-methyl-2-pentyl] phthalate were not resolved on any of the seven phases. The elution order was comparable on the Rtx-440, Rxi-XLB, Rtx-CLPesticides, and Rxi-5ms columns.
Table I: Predicted Elution Times for Regulated Phthalates on Various Restek GC Columns
Column: 30 m x 0.25 mm x 0.25 μm (0.20 μm for Rtx-CLPesticides2 column)
Constant linear velocity: 66.7 cm/sec
Oven: 200 °C (hold 0.5 min) to 330 °C (320 °C for Rtx-50 column) at 30 °C/min (hold 1 min)
| Peak # | Name | Listed | Retention Time (min) | CAS # | Purity | ||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Rtx-440 (cat. 12923) |
Rxi-XLB (cat. 13723) |
Rtx- CLPesticides (cat. 11123) |
Rxi-35Sil MS (cat. 13823) | Rtx-50 (cat. 10523) |
Rxi-5ms (cat. 13423) |
Rtx- CLPesticides2 (cat. 11323) |
|||||
| 1 | Dimethyl phthalate* | EPA 8061A, EPA Priority | 1.28 | 1.16 | 1.14 | 1.29 | 1.46 | 1.10 | 1.23 | 131-11-3 | Neat |
| 2 | Diethyl phthalate* | EPA 8061A, EPA Priority, EU | 1.54 | 1.39 | 1.33 | 1.55 | 1.73 | 1.30 | 1.47 | 84-66-2 | Neat |
| 3 | Benzyl benzoate | Internal Standard | 2.11 | 1.87 | 1.56 | 2.17 | 2.31 | 1.70 | 1.88 | 120-51-4 | Neat |
| 4 | Diisobutyl phthalate* | EPA 8061A | 2.25 | 2.04 | 1.88 | 2.21 | 2.34 | 1.91 | 2.10 | 84-69-5 | Neat |
| 5 | Di-n-butyl phthalate* | EPA 8061A, EPA Priority | 2.58 | 2.33 | 2.10 | 2.53 | 2.69 | 2.17 | 2.38 | 84-74-2 | Neat |
| 6 | Bis(2-methoxyethyl) phthalate* | EPA 8061A | 2.74 | 2.48 | 2.26 | 2.86 | 3.10 | 2.27 | 2.63 | 117-82-8 | Neat |
| 7 | Bis(4-methyl-2-pentyl) phthalate isomer 1* | EPA 8061A | 2.85 | 2.62 | 2.37 | 2.71 | 2.83 | 2.50 | 2.64 | 84-63-9 | Neat |
| 8 | Bis(4-methyl-2-pentyl) phthalate isomer 2* | EPA 8061A | 2.86 | 2.63 | 2.37 | 2.72 | 2.84 | 2.51 | 2.65 | 84-63-9 | Neat |
| 9 | Bis(2-ethoxyethyl) phthalate* | EPA 8061A | 3.08 | 2.80 | 2.51 | 3.13 | 3.33 | 2.59 | 2.90 | 605-54-9 | Neat |
| 10 | Di-n-pentyl phthalate* | EPA 8061A | 3.16 | 2.91 | 2.58 | 3.08 | 3.21 | 2.71 | 2.89 | 131-18-0 | Neat |
| 11 | Di-n-hexyl phthalate* | EPA 8061A | 3.73 | 3.46 | 3.07 | 3.61 | 3.69 | 3.25 | 3.42 | 84-75-3 | Neat |
| 12 | Butyl benzyl phthalate* | EPA 8061A, EPA Priority, EU | 3.85 | 3.56 | 3.12 | 3.93 | 4.13 | 3.30 | 3.63 | 85-68-7 | Neat |
| 13 | Hexyl 2-ethylhexyl phthalate | EPA 8061A | 3.98 | 3.72 | 3.29 | 3.83 | 3.92 | 3.52 | 3.66 | 75673-16-4 | Technical |
| 14 | Bis(2-butoxyethyl) phthalate* | EPA 8061A | 4.12 | 3.82 | 3.39 | 4.08 | 4.21 | 3.60 | 3.85 | 117-83-9 | Neat |
| 15 | Bis(2-ethylhexyl) phthalate* | EPA 8061A | 4.21 | 3.95 | 3.52 | 4.05 | 4.12 | 3.82 | 3.91 | 117-81-7 | Neat |
| 16 | Dicyclohexyl phthalate* | EPA 8061A, EPA Priority, EU | 4.33 | 4.04 | 3.55 | 4.42 | 4.58 | 3.78 | 4.08 | 84-61-7 | Neat |
| 17 | Di-n-octyl phthalate* | EPA 8061A, EPA Priority, EU | 4.76 | 4.50 | 3.97 | 4.59 | 4.62 | 4.24 | 4.39 | 117-84-0 | Neat |
| 18 | Diisononyl phthalate | EU | 5.10 | 4.84 | 4.23 | 4.84 | 4.84 | 4.50 | 4.64 | 68515-48-0 | Isomer mix |
| 19 | Diisodecyl phthalate | EU | 5.20 | 4.95** | 4.42 | 5.01 | 5.18 | 4.71 | 4.90 | 26761-40-0 | Isomer mix |
| 20 | Dinonyl phthalate* | EPA 8061A | 5.24 | 4.95** | 4.39 | 5.04 | 5.10 | 4.72 | 4.83 | 84-76-4 | Neat |
Note: Shading indicates coeluting peaks (Rs<1.5). For each column, different colors of shading indicate different coelution pairs.
*These compounds are contained in Restek’s EPA Method 8061A phthalates esters mixture (cat.# 33227).
**Peaks 19 and 20 are non-isobaric and can be separated using selected ion monitoring.
Differences in the elution orders were observed on Rxi-35Sil MS and Rtx-50 phases. Notably, the elution orders of four pairs of phthalates changed on the Rxi-35Sil MS phase, including bis(2-methoxyethyl) phthalate/bis(4-methyl-2-pentyl) phthalate isomers (peaks 6 and 7/8), bis(2-ethoxyethyl) phthalate/di-n-pentyl phthalate (peaks 9 and 10), butyl benzyl phthalate/hexyl-2-ethylhexyl phthalate (peaks 12 and 13), and bis(2-butoxyethyl) phthalate/bis(2-ethylhexyl) phthalate (peaks 14 and 15).
Rtx-440 and Rxi-XLB columns had the best overall separations under these conditions. Peaks that coeluted on other phases were well resolved on Rtx-440 and Rxi-XLB columns. Pairs that were not resolved on other phases include bis(2-ethylhexyl) phthalate and dicyclohexyl phthalate (peaks 15 and 16) on the Rxi-5ms column; bis(2-ethylhexyl) phthalate and butyl benzyl phthalate (peaks 15 and 12) on the Rtx-50 column; and bis(2-methoxyethyl) and bis[4-methyl-2-pentyl] phthalate (peaks 6 and 7,8) as well as bis(2-ethoxyethyl) phthalate and di-n-pentyl phthalate (peaks 9 and 10) on the Rtx-CLPesticides2 column. In technical grade isomer mixtures, it is possible to identify isomer groups, such as diisononyl phthalate and diisodecyl phthalate (peaks 18 and 19), but it is impossible to completely resolve each individual isomer within a group. Fortunately, unique extracted ions are available for identification and quantification, e.g., m/z 293 for diisononyl phthalate, and m/z 307 for diisodecyl phthalate (Figure 1).
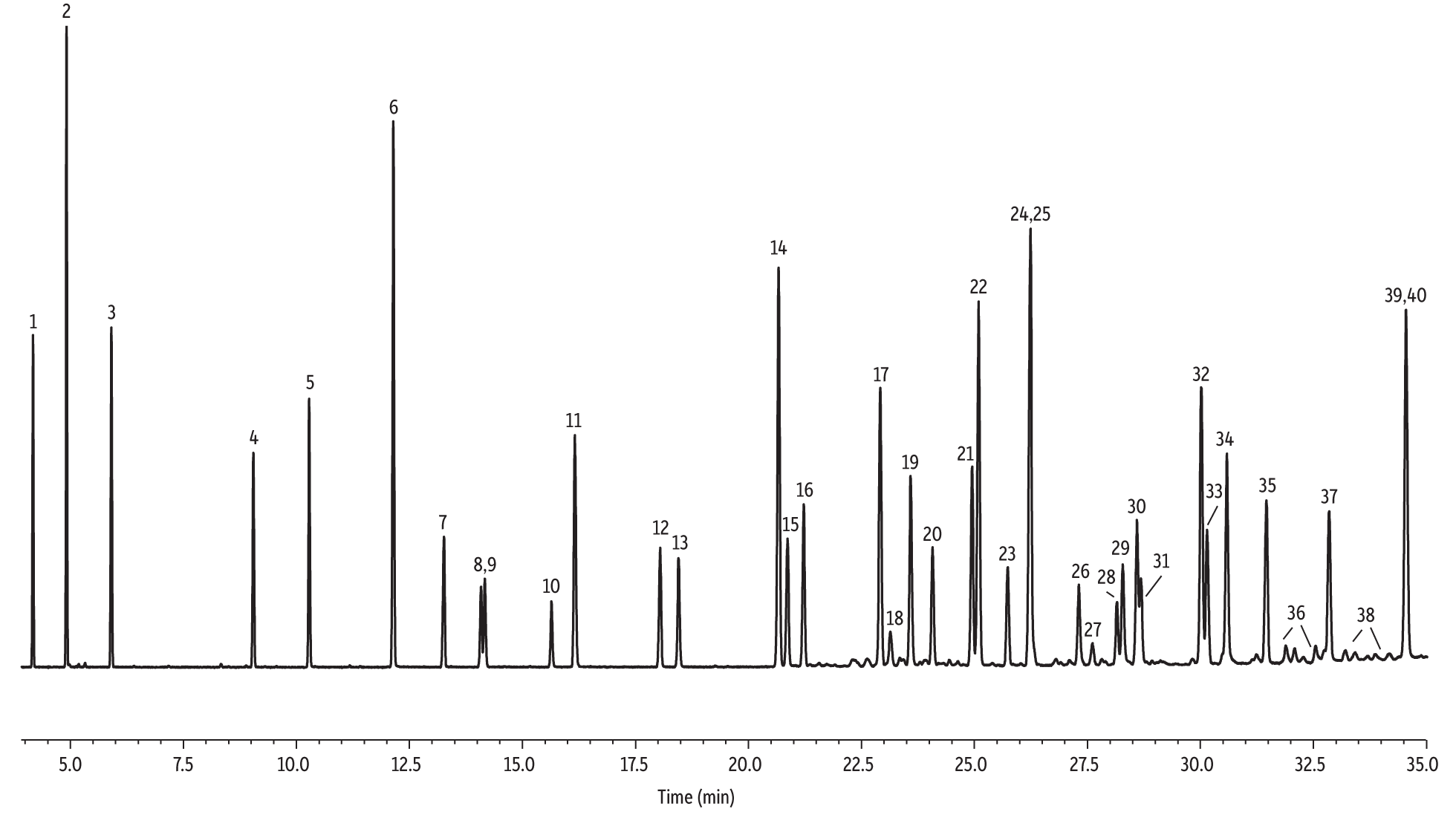
A more comprehensive comparison of the seven stationary phases was also conducted for the separation of 37 phthalates (a total number of 40 peaks, including three isomers) using the retention times predicted by the Pro EZGC ROM program (Table II). The GC parameters specified in Table III provided separation of 34 out of 40 peaks on both Rtx-440 and Rxi-XLB columns in less than 40 minutes and the two phases produced different coelutions. The chromatogram on the Rtx-440 column was collected and shown in Figure 2. For some pairs that were not baseline-resolved, the resolution is still adequate for qualitative analysis. There is no single set of conditions that is optimal for all phases. The program with the best overall results in terms of speed and the most peaks resolved was selected for this direct column comparison. Analysts can optimize conditions for their specific target lists using the Pro EZGC ROM program. Based on the overall analysis speed and high degree of target analyte separation, the Rtx-440 and Rxi-XLB columns are recommended for phthalate GC-MS analysis.
Table II: Predicted Elution Times for Phthalates (Extended List) on Various Restek GC Columns
Column: 30 m x 0.25 mm x 0.25 μm (0.20 μm for Rtx-CLPesticides2 column)
Constant linear velocity: 48 cm/sec
Oven: 150 °C (hold 0.8 min) to 200 °C at 5 °C/min to 275 °C at 3 °C/min (hold 2 min)
| Peak # | Name | Listed | Retention Time (min) | CAS # | Purity | ||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Rtx-440 (cat. 12923) |
Rxi-XLB (cat. 13723) |
Rxi-5ms (cat. 13423) | Rtx-50 (cat. 10523) | Rxi-35Sil MS (cat. 13823) | Rtx- CLPesticides (cat. 11123) |
Rtx- CLPesticides2 (cat. 11323) |
|||||
| 1 | Dimethyl phthalate* | EPA 8061A, EPA Priority | 4.606 | 3.924 | 3.294 | 5.912 | 4.902 | 3.75 | 4.334 | 131-11-3 | Neat |
| 2 | Dimethyl isophthalate | — | 5.491 | 4.690 | 3.85 | 6.35 | 5.498 | 4.174 | 4.793 | 1459-93-4 | Neat |
| 3 | Diethyl phthalate* | EPA 8061A, EPA Priority, EU | 6.537 | 5.642 | 4.762 | 7.809 | 6.785 | 5.24 | 6.106 | 84-66-2 | Neat |
| 4 | Benzyl benzoate | Internal Standard | 9.931 | 8.667 | N/A | 11.099 | N/A | 6.725 | 8.583 | 120-51-4 | Neat |
| 5 | Diisobutyl phthalate* | EPA 8061A | 11.185 | 10.029 | 8.817 | 11.817 | 11.008 | 9.101 | 10.333 | 84-69-5 | Neat |
| 6 | Di-n-butyl phthalate* | EPA 8061A, EPA Priority | 13.152 | 11.850 | 10.405 | 14.031 | 13.094 | 10.481 | 12.029 | 84-74-2 | Neat |
| 7 | Bis(2-methoxyethyl) phthalate* | EPA 8061A | 14.343 | 12.784 | 11.045 | 17.095 | 15.424 | 11.54 | 13.725 | 117-82-8 | Neat |
| 8 | Bis(4-methyl-2-pentyl) phthalate isomer 1* | EPA 8061A | 15.192 | 13.754 | 12.47 | 15.184 | 14.454 | 12.166 | 13.825 | 84-63-9 | Neat |
| 9 | Bis(4-methyl-2-pentyl) phthalate isomer-2* | EPA 8061A | 15.350 | 13.828 | 12.55 | 15.277 | 14.542 | 12.233 | 13.906 | 84-63-9 | Neat |
| 10 | Bis(2-ethoxyethyl) phthalate* | EPA 8061A | 16.910 | 15.132 | 13.199 | 19.063 | 17.59 | 13.186 | 15.875 | 605-54-9 | Neat |
| 11 | Di-n-pentyl phthalate* | EPA 8061A | 17.454 | 15.880 | 13.856 | 17.974 | 17.128 | 13.588 | 15.768 | 131-18-0 | Neat |
| 12 | Butyl cyclohexyl phthalate | — | 19.452 | 17.689 | 15.478 | 21.19 | 19.843 | 14.979 | 17.96 | 84-64-0 | Technical |
| 13 | Butyl 2-ethylhexyl phthalate | — | 19.823 | 18.172 | 16.174 | 20.062 | 19.238 | 15.566 | 17.958 | 85-69-8 | Technical |
| 14 | Di-n-hexyl phthalate* | EPA 8061A | 22.138 | 20.279 | 17.984 | 22.152 | 21.469 | 17.215 | 19.829 | 84-75-3 | Neat |
| 15 | Butyl octyl phthalate | — | 22.338 | 20.557 | 18.136 | 22.37 | 21.668 | 17.344 | 20.009 | 84-78-6 | Technical |
| 16 | Butyl benzyl phthalate* | EPA 8061A, EPA Priority, EU | 22.799 | 20.783 | 18.029 | 25.365 | 23.782 | 17.384 | 21.128 | 85-68-7 | Neat |
| 17 | Hexyl 2-ethylhexyl phthalate | EPA 8061A | 24.404 | 22.668 | 20.266 | 24.110 | 23.500 | 19.126 | 22.049 | 75673-16-4 | Technical |
| 18 | Butyl isodecyl phthalate | — | 24.632 | 22.793 | 20.392 | 24.220 | 23.685 | 19.424 | 22.22 | 42343-36-2 | Technical |
| 19 | Bis(2-ethylhexyl)hexahydro phthalate | — | 25.066 | 23.389 | 21.254 | 23.089 | 23.063 | 19.142 | 21.961 | 84-71-9 | Neat |
| 20 | Bis(2-n-butoxyethyl) phthalate* | EPA 8061A | 25.601 | 23.563 | 20.930 | 26.746 | 25.647 | 19.849 | 23.533 | 117-83-9 | Neat |
| 21 | Dicyclohexyl phthalate* | EPA 8061A, EPA Priority, EU | 26.651 | 24.495 | 21.771 | 28.989 | 27.671 | 20.530 | 24.792 | 84-61-7 | Neat |
| 22 | Bis(2-ethylhexyl) phthalate* | EPA 8061A | 26.692 | 24.845 | 22.585 | 25.903 | 25.458 | 21.135 | 24.048 | 117-81-7 | Neat |
| 23 | Butyl-n-decyl phthalate | — | 27.362 | 25.268 | 22.657 | 26.888 | 26.410 | 21.404 | 24.471 | 89-19-0 | Technical |
| 24 | Diphenyl phthalate | — | 27.987 | 25.712 | 22.372 | 32.277 | 30.170 | 21.614 | 26.473 | 84-62-8 | Neat |
| 25 | Bis(4-methylcyclohexyl) phthalate isomer 1 | — | 28.003 | 25.922 | 23.016 | 29.547 | 28.476 | 21.677 | 25.923 | 59-43-8 | Isomer mix |
| 26 | Bis(4-methylcyclohexyl) phthalate isomer 2 | — | 29.002 | 26.993 | 23.816 | 30.345 | 29.400 | 22.604 | 26.739 | 59-43-8 | Isomer mix |
| 27 | Hexyl isodecyl phthalate | — | 29.176 | 27.271 | 24.523 | 28.189 | 27.965 | 23.224 | 26.336 | 61702-81-6 | Technical |
| 28 | Benzyl 2-ethylhexyl phthalate | — | 29.791 | 27.781 | 24.747 | 31.498 | 30.216 | 23.219 | 27.594 | 27215-22-1 | Technical |
| 29 | Bis(4-methylcyclohexyl) phthalate isomer-3 | — | 29.964 | 28.034 | 24.617 | 31.189 | 30.285 | 23.498 | 27.559 | 59-43-8 | Isomer mix |
| 30 | Bis(2-ethylhexyl) isophthalate | — | 30.132 | 28.037 | 25.684 | 28.133 | 28.243 | 23.907 | 26.648 | 137-89-3 | Neat |
| 31 | Bis(2-(ethoxyethoxy)ethyl) phthalate | — | 30.233 | 28.434 | 24.879 | 32.942 | 31.252 | 23.995 | 28.681 | 117-85-1 | Technical |
| 32 | Di-n-octyl phthalate* | EPA 8061A, EPA Priority, EU | 31.562 | 29.626 | 26.796 | 30.475 | 30.328 | 24.915 | 28.455 | 117-84-0 | Neat |
| 33 | n-Hexyl decyl phthalate | — | 31.680 | 29.748 | 26.878 | 30.788 | 30.450 | 24.994 | 28.566 | 25724-58-7 | Technical |
| 34 | Diphenyl isophthalate | — | 32.362 | 29.850 | N/A | 34.707 | 32.396 | 25.114 | 29.437 | 744-45-6 | Neat |
| 35 | Dibenzyl phthalate | — | 33.234 | 30.725 | 27.141 | 37.396 | 35.372 | 25.501 | 31.359 | 523-31-9 | Neat |
| 36 | Diisononyl phthalate | EU | 33.684 | 31.802 | 28.779 | 32.500 | 32.708 | 27.391 | 30.811 | 68515-48-0 | Isomer mix |
| 37 | Di-n-octyl isophthalate | — | 34.483 | 32.463 | 29.168 | 32.035 | N/A | 27.223 | 30.388 | 4654-18-6 | Neat |
| 38 | Diisodecyl phthalate | EU | 35.775 | 33.792 | 30.876 | 35.041 | N/A | 29.11 | 32.169 | 26761-40-0 | Isomer mix |
| 39 | Dinonyl phthalate* | EPA 8061A | 36.159 | 34.103 | 30.994 | 34.609 | 34.705 | 28.867 | 32.604 | 84-76-4 | Neat |
| 40 | n-Octyl-n-decyl phthalate | — | 36.182 | 34.170 | 30.961 | 34.664 | 34.7 | 28.861 | 32.628 | 119-07-3 | Technical |
Note: Shading indicates coeluting peaks (Rs<1.5). For each column, different colors of shading indicate different coelution pairs.
*These compounds are contained in Restek’s EPA Method 8061A phthalates esters mixture (cat.# 33227).
Table III: GC-MS Parameters
| Parameters | Values for EPA EU List | Values for Extended List |
|---|---|---|
| Inlet temperature (°C) | 280 | 280 |
| Injection volume (µL) | 1.0 | 1.0 |
| Liner | Restek Premium 3.5 mm Precision liner w/ wool (cat.# 23320.1) | Restek Premium 3.5 mm Precision liner w/ wool (cat.# 23320.1) |
| Oven temperature program | 200 °C (hold 0.5 min) to 330 °C* at 30 °C/min (hold 1 min) |
150 °C (hold 0.8 min) to 200 °C at 5 °C/min to 275 °C at 3 °C/min (hold 2 min) |
| Carrier gas: He control mode | Constant linear velocity: 66.7 cm/sec @ 200 °C (3 mL/min**) |
Constant linear velocity: 48 cm/sec @ 150 °C (1.6 mL/min) |
| Split ratio | 20:1 | 20:1 |
| Detector | MS | MS |
| Mode: | Full Scan (59–400) | Full Scan (59–400) |
| Transfer line temp.: | 300 °C | 300 °C |
| Scan event time | 0.1 sec | 0.1 sec |
| Analyzer type: | Quadrupole | Quadrupole |
| Source temp.: | 280 °C | 280 °C |
| Solvent delay time: | 0.9 min | 2.5 min |
| Tune type: | PFTBA | PFTBA |
| Ionization mode: | EI | EI |
*320 °C for Rtx-50 column
**3 mL/min may be too high for some instruments. Consult your instrument operations manual prior to programming.
Although GC-MS is generally preferred because it provides more definitive information, phthalate analysis can also be successfully accomplished by GC-ECD. EPA 8061A is a method used to identify and quantify phthalates in aqueous and solid matrices using a parallel column configuration and dual electron capture detectors [8]. Rtx-440 and Rxi-35Sil MS columns are ideal as a parallel dual column set. The Pro EZGC software provided rapid analysis conditions using the Rtx-440 column, and the Rxi-35Sil MS column served as an excellent confirmation column, due to the elution order changes that were observed. GC-ECD conditions can be easily translated from the GC-MS methods shown in Table III using Restek’s free online EZGC method translator; alternatively, optimized GC-ECD instrument parameters and example chromatograms are available in this blog [9].
Conclusion
The seven GC columns most commonly used for phthalate analysis were directly compared using the Pro EZGC program, which provided flexibility and ease in GC optimization. The superior selectivity and high efficiency of the Rtx-440 and Rxi-XLB columns resulted in fast analysis times for both the regulated phthalates and the extended list. With good resolution, high maximum operating temperatures (340 °C for Rtx-440 and 360 °C for Rxi-XLB [Table IV]), and minimum phase bleed, the Rtx-440 and Rxi-XLB columns are the preferred choices for phthalate GC-MS analysis. A dual column set of Rtx-440 and Rxi-35Sil MS columns is recommended if GC-ECD is used instead of GC-MS.
Table IV: Maximum Temperatures
| Rtx-440 (cat. 12923) |
Rxi-XLB (cat. 13723) |
Rxi-5ms (cat. 13423) |
Rtx-50 (cat. 10523) |
Rxi-35Sil MS (cat. 13823) |
Rtx-CLPesticides (cat. 11123) |
Rtx-CLPesticides2 (cat. 11323) |
|
|---|---|---|---|---|---|---|---|
| Maximum Temperature (°C) | 340 | 360 | 350 | 320 | 360 | 340 | 340 |
Acknowledgements
The authors would like to thank Shimadzu Corporation for their consultation.
References
- H. Choi, J. Kim, Y. Im, S. Lee, Y. Kim, The association between some endocrine disruptors and hypospadias in biological samples. J. Environ. Sci. Health, Part A: Toxic/Hazard. Subst. Environ. Eng. 47 (13) (2012) 2173–2179. http://www.ncbi.nlm.nih.gov/pubmed/22871016
- N. Nassar, P. Abeywardana, A. Barker, C. Bower, Parental occupational exposure to potential endocrine disrupting chemicals and risk of hypospadias in infants. Occup. Environ. Med. 67 (9) (2010) 585–589. http://www.ncbi.nlm.nih.gov/pubmed/19939854
- L. Trasande, S. Sathyanarayana, A.J. Spanier, H. Trachtman, T.M. Attina, E.M. Urbina, Urinary phthalates are associated with higher blood pressure in childhood. J. Pediatr. 163 (3) (2013) 747–753e1. http://www.ncbi.nlm.nih.gov/pmc/articles/PMC4074773/
- E.F. Werner, J.M. Braun, K. Yolton, J.C. Khoury, B.P. Lanphear, The association between maternal urinary phthalate concentrations and blood pressure in pregnancy: The HOME Study. Environ. Health 14 (2015) 75. http://www.ehjournal.net/content/14/1/75
- J.J. Jaakkola, T.L. Knight, The role of exposure to phthalates from polyvinyl chloride products in the development of asthma and allergies: A systematic review and meta-analysis. Environ. Health Perspect. 116 (7) (2008) 845–853. http://www.ncbi.nlm.nih.gov/pmc/articles/PMC2453150/
- E.E. Hatch, J.W. Nelson, R.W. Stahlhut, T.F. Webster, Association of endocrine disruptors and obesity: Perspectives from epidemiological studies. Int. J. Androl. 33 (2) (2010) 324–332. http://www.ncbi.nlm.nih.gov/pubmed/20113374
- S. Net, A. Delmont, R. Sempere, A. Paluselli, B. Ouddane, Reliable quantification of phthalates in environmental matrices (air, water, sludge, sediment and soil): A review. Sci. Total Environ. 515-516 (2015) 162–180. http://www.ncbi.nlm.nih.gov/pubmed/25723871
- U.S. Environmental Protection Agency, Method 8061A, Phthalate Esters by Gas Chromatography with Electron Capture Detection (GC/ECD), Rev. 1, December 1996. https://www.epa.gov/sites/production/files/2015-12/documents/8061a.pdf
- D. Li, Phthalate determination by dual column set in eight minutes, ChromaBLOGraphy, Restek Corporation, 2015. http://blog.restek.com/?p=17388
Figure 1: EPA- and EU-listed phthalates and internal standard (benzyl benzoate) are shown under full scan mode and selected ion scan mode (m/z 293 and m/z 307) on seven different GC stationary phases (see Table III for conditions).
Rtx-440
| Peaks | |
|---|---|
| 1. | Dimethyl phthalate |
| 2. | Diethyl phthalate |
| 3. | Benzyl benzoate |
| 4. | Diisobutyl phthalate |
| 5. | Di-n-butyl phthalate |
| 6. | Bis(2-methoxyethyl) phthalate |
| 7. | Bis[4-methyl-2-pentyl] phthalate isomer 1 |
| 8. | Bis[4-methyl-2-pentyl] phthalate isomer 2 |
| 9. | Bis(2-ethoxyethyl) phthalate |
| 10. | Di-n-pentyl phthalate |
Rxi-XLB
Rtx-CLPesticides
Rtx-CLPesticides2
Figure 2: Target compounds on the extended phthalate list (50 µg/mL) are separated under full scan mode on an Rtx-440 column (see Table III for conditions).