Accurately Analyze Metal-Sensitive Compounds with Restek’s New Inert LC Columns
Our new inert LC column technology helps labs improve their analysis of metal-sensitive compounds. A premium inert coating applied to the stainless-steel surface of our LC columns reduces nonspecific binding of chelating analytes, enabling sensitive analysis and smooth integration of peaks. Combined with Restek’s selective stationary phases, these new inert LC columns are ideal for the analysis of metal-sensitive compounds, such as organophosphorus pesticides and mycotoxins.
Restek’s inert LC columns can provide four key benefits:
- Improved peak shape without passivation or mobile phase additives.
- Increased response and analyte recovery, allowing lower detection limits.
- High accuracy and throughput with less variability.
- Less time-consuming conditioning and complicated passivation required.
Exceptional Inertness Brings Exceptional Performance
Analyzing compounds that have nonspecific adsorption (NSA) or nonspecific binding (NSB) to metal surfaces in LC columns have historically been a challenge. Poor peak shape and sensitivity are key indicators that polar, usually acidic compounds are interacting with the metal surfaces in the column, causing poor data quality. Our premium inert column technology is designed to eliminate NSA and NSB of active analytes to the column hardware, giving analysts greater confidence in their data when working with metal-sensitive analytes.
Pesticides
Pesticide panels benefit from the use of inert LC columns as they contain a wide variety of compounds. Phosphorylated, acidic, polar compounds, and/or metal chelating species, such as organophosphate pesticides, are reactive to the metal surfaces inside of the analytical column. Our new inert LC columns solve that problem easily to improve the overall performance of your pesticide panel.
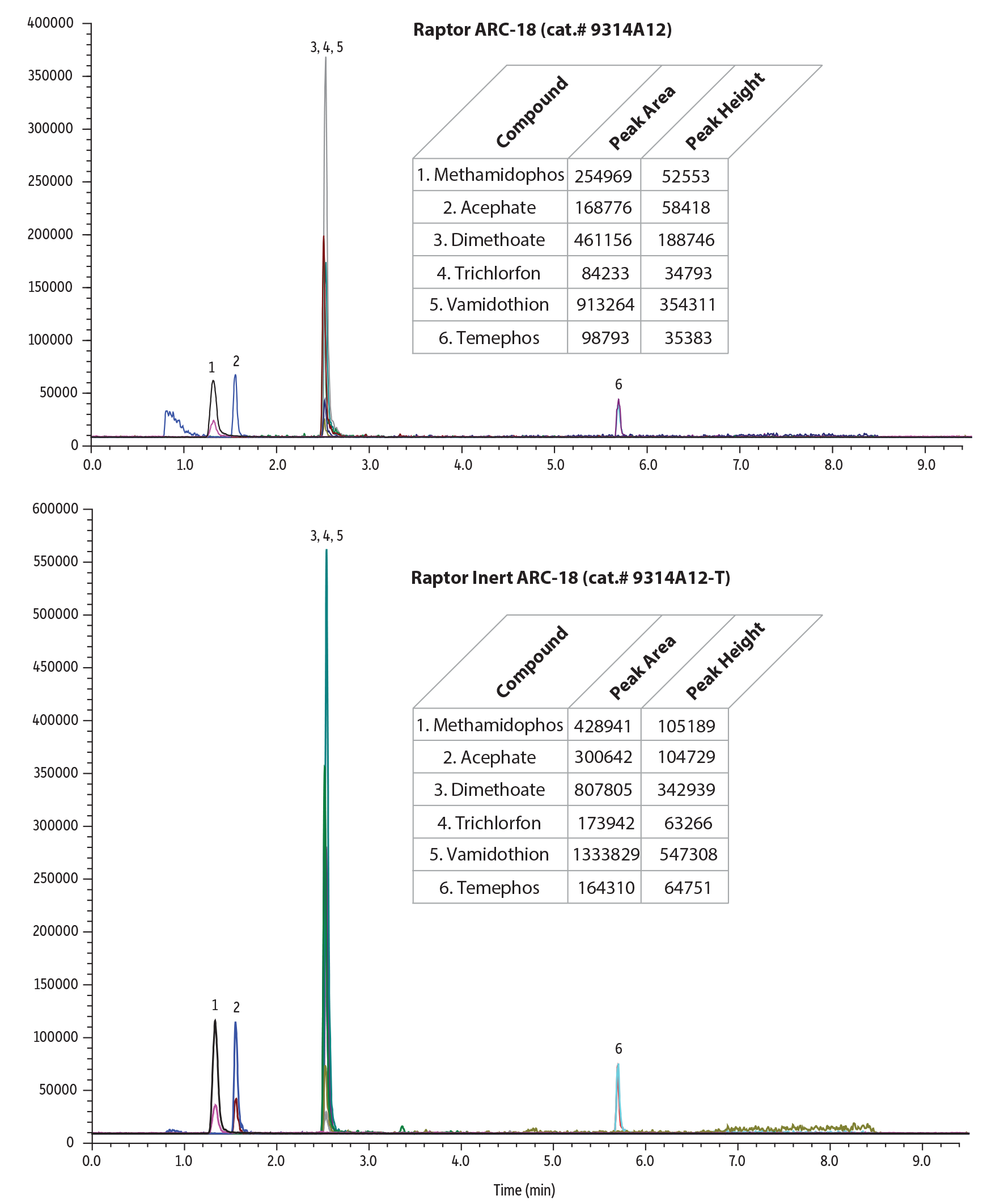
Figure 1: Our new Raptor inert ARC-18 columns offer increased sensitivity, recoveries, and lower detection limits without any column preconditioning.
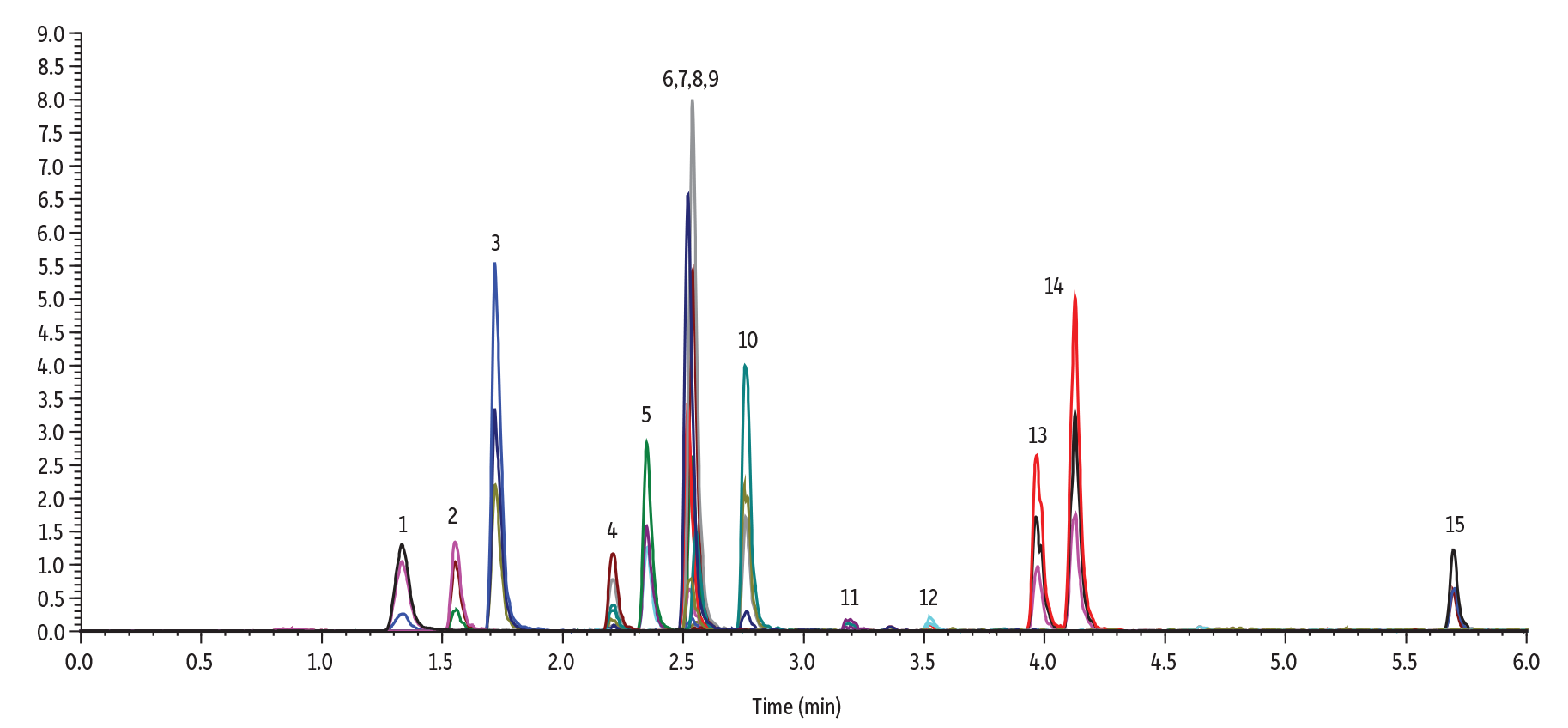
| Peaks | tR (min) | Precursor Ion | Product Ion 1 | Product Ion 2 | Peak Area | Peak Height | |
|---|---|---|---|---|---|---|---|
| 1. | Methamidophos | 1.33 | 142.0 | 94.0 | 125.1 | 428941 | 105189 |
| 2. | Acephate | 1.55 | 184.0 | 143.0 | 48.9 | 300642 | 104729 |
| 3. | Omethoate | 1.72 | 214.0 | 125.0 | 182.9 | 892008 | 337690 |
| 4. | Monocrotophos | 2.21 | 224.1 | 127.0 | 193.1 | 215810 | 78425 |
| 5. | Dicrotophos | 2.35 | 238.1 | 112.1 | 72.0 | 404916 | 159292 |
| 6. | Dimethoate | 2.52 | 230.0 | 125.0 | 199.0 | 807805 | 342939 |
| 7. | Trichlorfon | 2.53 | 257.0 | 108.9 | 220.8 | 173942 | 63266 |
| 8. | Vamidothion | 2.54 | 288.0 | 146.0 | 118.0 | 1333829 | 547308 |
| 9. | Mevinphos isomer 1 | 2.55 | 241.9 | 126.9 | 192.9 | 311274 | 129961 |
| 10. | Mevinphos isomer 2 | 2.76 | 241.9 | 126.9 | 192.9 | 74030 | 29802 |
| 11. | Carbaryl | 3.18 | 202.1 | 145.0 | 127.0 | 39671 | 11924 |
| 12. | Isocarbophos | 3.52 | 291.1 | 231.1 | 121.1 | 33294 | 11941 |
| 13. | Dimethomorph isomer 1 | 3.96 | 388.2 | 300.9 | 165.1 | 511766 | 172977 |
| 14. | Dimethomorph isomer 2 | 4.13 | 388.2 | 300.9 | 165.1 | 877031 | 328826 |
| 15. | Temephos | 5.70 | 467.1 | 124.9 | 418.9 | 164310 | 64751 |
| Column | Raptor Inert ARC-18 (cat.# 9314A12-T) | ||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Dimensions: | 100 mm x 2.1 mm ID | ||||||||||||||||||||||||||||||||
| Particle Size: | 2.7 µm | ||||||||||||||||||||||||||||||||
| Pore Size: | 90 Å | ||||||||||||||||||||||||||||||||
| Temp.: | 50 °C | ||||||||||||||||||||||||||||||||
| Standard/Sample | LC multiresidue pesticide standard #1 (cat.# 31972) | ||||||||||||||||||||||||||||||||
| Diluent: | Water, 0.1% formic acid | ||||||||||||||||||||||||||||||||
| Conc.: | 1 ng/mL | ||||||||||||||||||||||||||||||||
| Inj. Vol.: | 5 µL | ||||||||||||||||||||||||||||||||
| Mobile Phase | |||||||||||||||||||||||||||||||||
| A: | Water, 2 mM ammonium formate, 0.1% formic acid | ||||||||||||||||||||||||||||||||
| B: | Methanol, 2 mM ammonium formate, 0.1% formic acid | ||||||||||||||||||||||||||||||||
|
| Detector | Shimadzu LCMS-8060 |
|---|---|
| Ion Mode: | ESI+ |
| Mode: | MRM |
| Instrument | Shimadzu Nexera X2 |
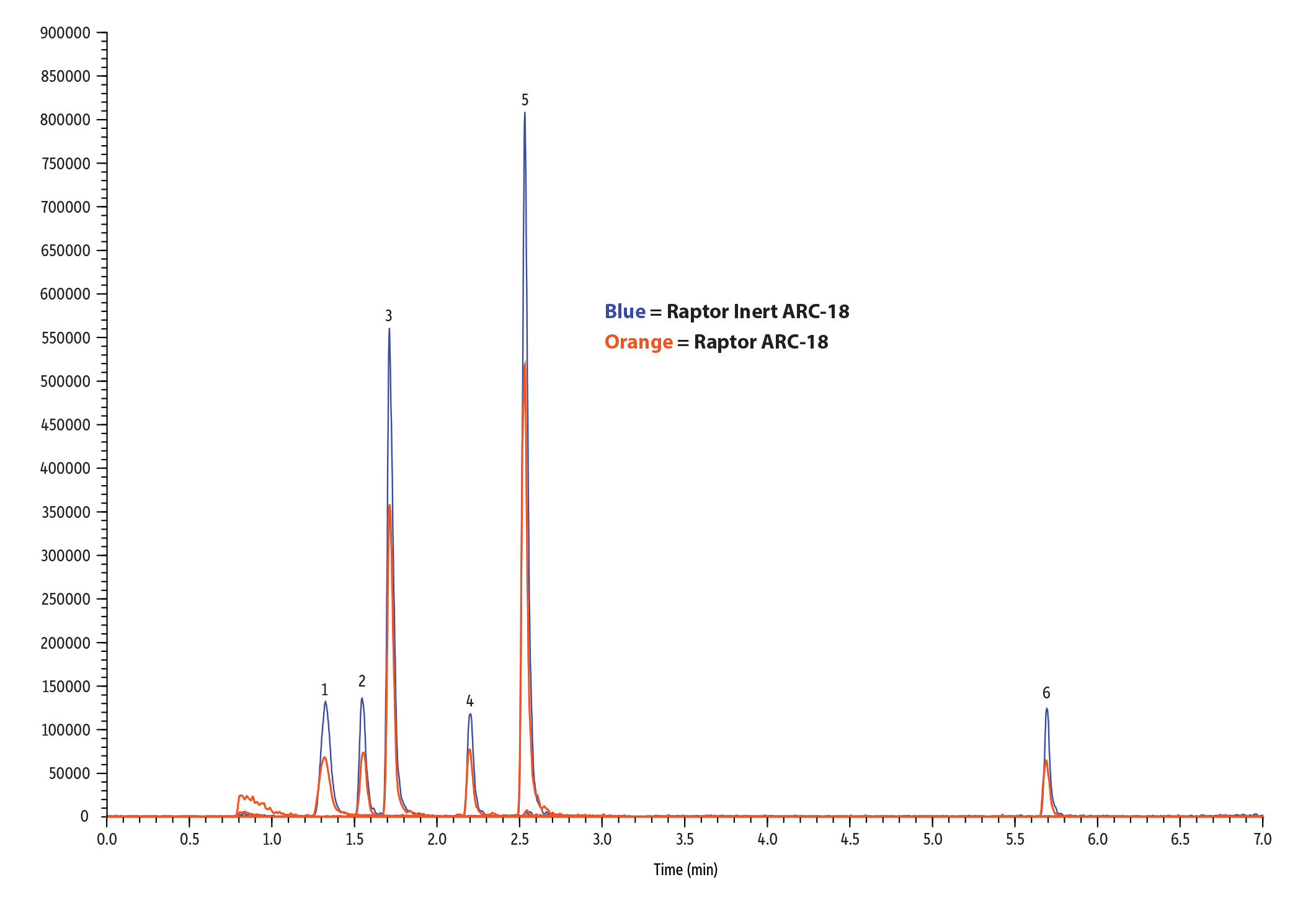
Figure 2: Compared to a conventional, stainless-steel column, our new Raptor inert ARC-18 columns provide exceptional analyte recoveries.
Figure 3: Peak area and peak height are greatly improved for the analysis of pesticides.
Table I: Restek's new inert columns showed up to 2X improvement in peak area and peak height over stainless-steel columns in this analysis of pesticides.
| Compound | Peak Area | Peak Height | ||||
| Stainless Steel | Inert | Areas Ratio (Inert/Stainless Steel) | Stainless Steel | Inert | Height Ratio (Inert/Stainless Steel) | |
| Methamidophos | 254969 | 428941 | 1.68 | 52553 | 105189 | 2.00 |
| Acephate | 168776 | 300642 | 1.78 | 58418 | 104729 | 1.79 |
| Omethoate | 579502 | 892008 | 1.54 | 216157 | 337690 | 1.56 |
| Monocrotophos | 140095 | 215810 | 1.54 | 51402 | 78425 | 1.53 |
| Dicrotophos | 340978 | 404916 | 1.19 | 135380 | 159292 | 1.18 |
| Dimethoate | 461156 | 807805 | 1.75 | 188746 | 342939 | 1.82 |
| Trichlorfon | 84233 | 173942 | 2.07 | 34793 | 63266 | 1.82 |
| Vamidothion | 913264 | 1333829 | 1.46 | 354311 | 547308 | 1.54 |
| Mevinphos isomer 1 | 213632 | 311274 | 1.46 | 82105 | 129961 | 1.58 |
| Mevinphos isomer 2 | 56093 | 74030 | 1.32 | 29070 | 29802 | 1.03 |
| Carbaryl | 43590 | 39671 | 0.91 | 14563 | 11924 | 0.82 |
| Isocarbophos | 21587 | 33294 | 1.54 | 9062 | 11941 | 1.32 |
| Dimethomorph isomer 1 | 462425 | 511766 | 1.11 | 166990 | 172977 | 1.04 |
| Dimethomorph isomer 2 | 896109 | 877031 | 0.98 | 311657 | 328826 | 1.06 |
| Temephos | 98793 | 164310 | 1.66 | 35383 | 64751 | 1.83 |
Mycotoxins
Mycotoxins analysis can be challenging and often requires a great deal of column conditioning and equilibration to achieve acceptable peaks. This is due to the reactive nature of the compounds, which contain acidic, polar, or otherwise metal chelating groups. Our new inert column hardware, combined with our stationary phases, helps simplify methods and improve the response and peak shape of these compounds.
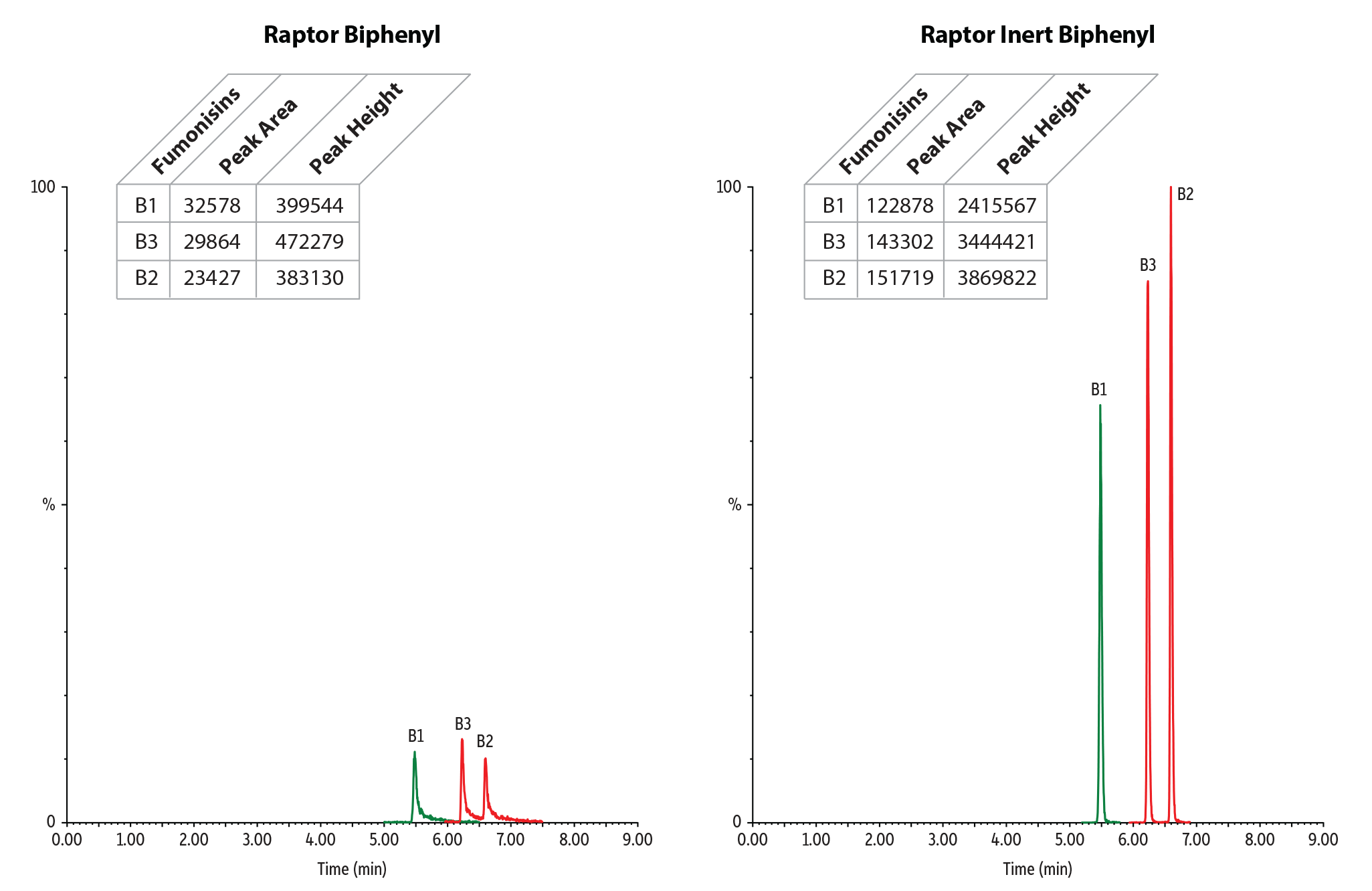
Figure 4: Achieve excellent peak shape without additional acid passivation or mobile phase additives when analyzing mycotoxins with our new Raptor inert biphenyl columns.
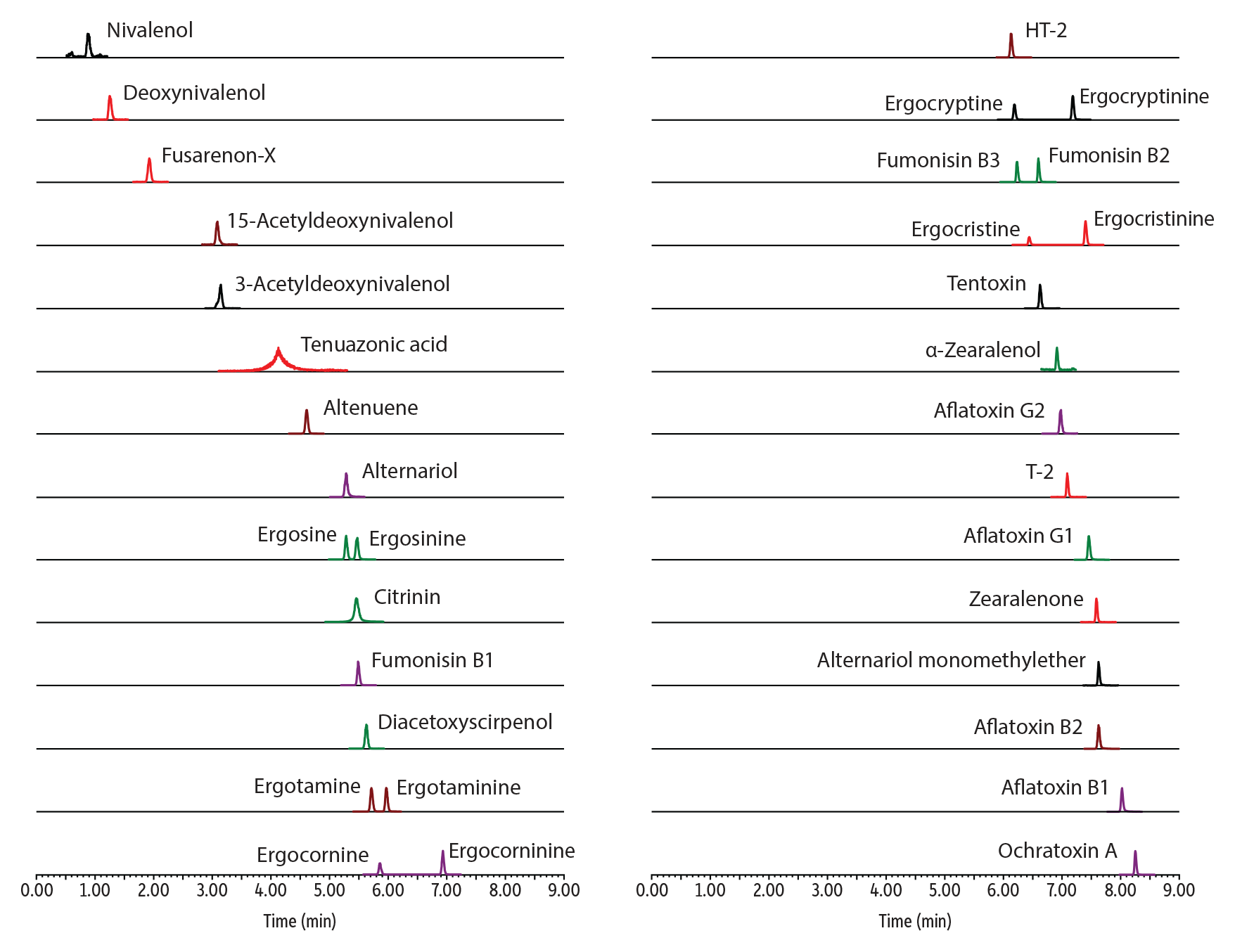
| Peaks | tR (min) | Conc. (ng/mL) | Precursor Ion | Product Ion | Peak Area | Peak Height | |
|---|---|---|---|---|---|---|---|
| 1. | Nivalenol | 0.88 | 10 | 295.1 | 137.1 | 4182 | 64495 |
| 2. | Deoxynivalenol | 1.25 | 10 | 297.2 | 231.0 | 17346 | 281906 |
| 3. | Fusarenon-X | 1.92 | 10 | 355.1 | 137.1 | 7668 | 121790 |
| 4. | 15-Acetyldeoxynivalenol | 3.08 | 10 | 339.2 | 137.1 | 31369 | 517570 |
| 5. | 3-Acetyldeoxynivalenol | 3.14 | 10 | 339.2 | 213.1 | 22613 | 296396 |
| 6. | Tenuazonic acid | 4.11 | 10 | 198.1 | 125.0 | 47828 | 197658 |
| 7. | Altenuene | 4.60 | 10 | 293.2 | 257.1 | 113850 | 2059699 |
| 8. | Alternariol | 5.27 | 10 | 259.0 | 185.1 | 73272 | 1302192 |
| 9. | Ergosine | 5.28 | 10 | 548.4 | 208.1 | 486620 | 9366601 |
| 10. | Citrinin | 5.46 | 10 | 251.2 | 233.1 | 1007880 | 9828889 |
| 11. | Ergosinine | 5.46 | 10 | 548.4 | 208.1 | 496734 | 8740527 |
| 12. | Fumonisin B1 | 5.48 | 10 | 722.5 | 352.3 | 122878 | 2415567 |
| 13. | Diacetoxyscirpenol | 5.62 | 10 | 384.2 | 247.1 | 68139 | 1208825 |
| 14. | Ergotamine | 5.71 | 10 | 582.4 | 223.2 | 493003 | 9274155 |
| 15. | Ergocornine | 5.85 | 10 | 562.4 | 268.2 | 387025 | 7732744 |
| 16. | Ergotaminine | 5.96 | 10 | 582.4 | 223.2 | 462119 | 9237991 |
| 17. | HT-2 | 6.13 | 10 | 447.2 | 345.1 | 15221 | 323765 |
| 18. | Ergocryptine | 6.19 | 10 | 576.4 | 268.2 | 522204 | 11360838 |
| 19. | Fumonisin B3 | 6.23 | 10 | 706.4 | 336.2 | 143302 | 3444421 |
| 20. | Ergocristine | 6.44 | 10 | 610.4 | 223.2 | 195562 | 4450058 |
| 21. | Fumonisin B2 | 6.59 | 10 | 706.4 | 336.2 | 151719 | 3869822 |
| 22. | Tentoxin | 6.62 | 10 | 415.2 | 312.2 | 95175 | 2131906 |
| 23. | α-Zearalenol | 6.91 | 10 | 303.1 | 285.1 | 30224 | 702420 |
| 24. | Ergocorninine | 6.93 | 10 | 562.4 | 268.2 | 704029 | 14389283 |
| 25. | Aflatoxin G2 | 6.97 | 10 | 331.2 | 189.0 | 262824 | 5274353 |
| 26. | T-2 | 7.09 | 10 | 489.2 | 387.1 | 56535 | 1394735 |
| 27. | Ergocryptinine | 7.18 | 10 | 576.4 | 268.2 | 778972 | 16765348 |
| 28. | Ergocristinine | 7.40 | 10 | 610.4 | 223.2 | 1583053 | 32975663 |
| 29. | Aflatoxin G1 | 7.45 | 10 | 329.1 | 199.7 | 304389 | 6102959 |
| 30. | Zearalenone | 7.59 | 10 | 319.2 | 283.1 | 37162 | 927455 |
| 31. | Alternariol monomethylether | 7.62 | 10 | 273.0 | 199.1 | 31024 | 640689 |
| 32. | Aflatoxin B2 | 7.63 | 10 | 315.1 | 287.0 | 295648 | 5724754 |
| 33. | Aflatoxin B1 | 8.02 | 10 | 313.2 | 241.1 | 223520 | 4425821 |
| 34. | Ochratoxin A | 8.25 | 10 | 404.1 | 239.0 | 190060 | 4411953 |
| Column | Raptor Inert Biphenyl (cat.# 9309A12-T) | ||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Dimensions: | 100 mm x 2.1 mm ID | ||||||||||||||||||||||||
| Particle Size: | 2.7 µm | ||||||||||||||||||||||||
| Pore Size: | 90 Å | ||||||||||||||||||||||||
| Temp.: | 60 °C | ||||||||||||||||||||||||
| Standard/Sample | Aflatoxins standard (cat.# 34121) | ||||||||||||||||||||||||
| Ochratoxin A standard (cat.# 34122) | |||||||||||||||||||||||||
| Diluent: | 50:50 Water:methanol | ||||||||||||||||||||||||
| Conc.: | 10 ng/mL | ||||||||||||||||||||||||
| Inj. Vol.: | 5 µL | ||||||||||||||||||||||||
| Mobile Phase | |||||||||||||||||||||||||
| A: | Water, 0.05% formic acid | ||||||||||||||||||||||||
| B: | Methanol, 0.05% formic acid | ||||||||||||||||||||||||
| |||||||||||||||||||||||||
| Max Pressure: | 440 bar |
| Detector | Waters Xevo TQ-S |
|---|---|
| Ion Mode: | ESI+ |
| Mode: | MRM |
| Instrument | Waters ACQUITY UPLC I-Class |
| Notes |
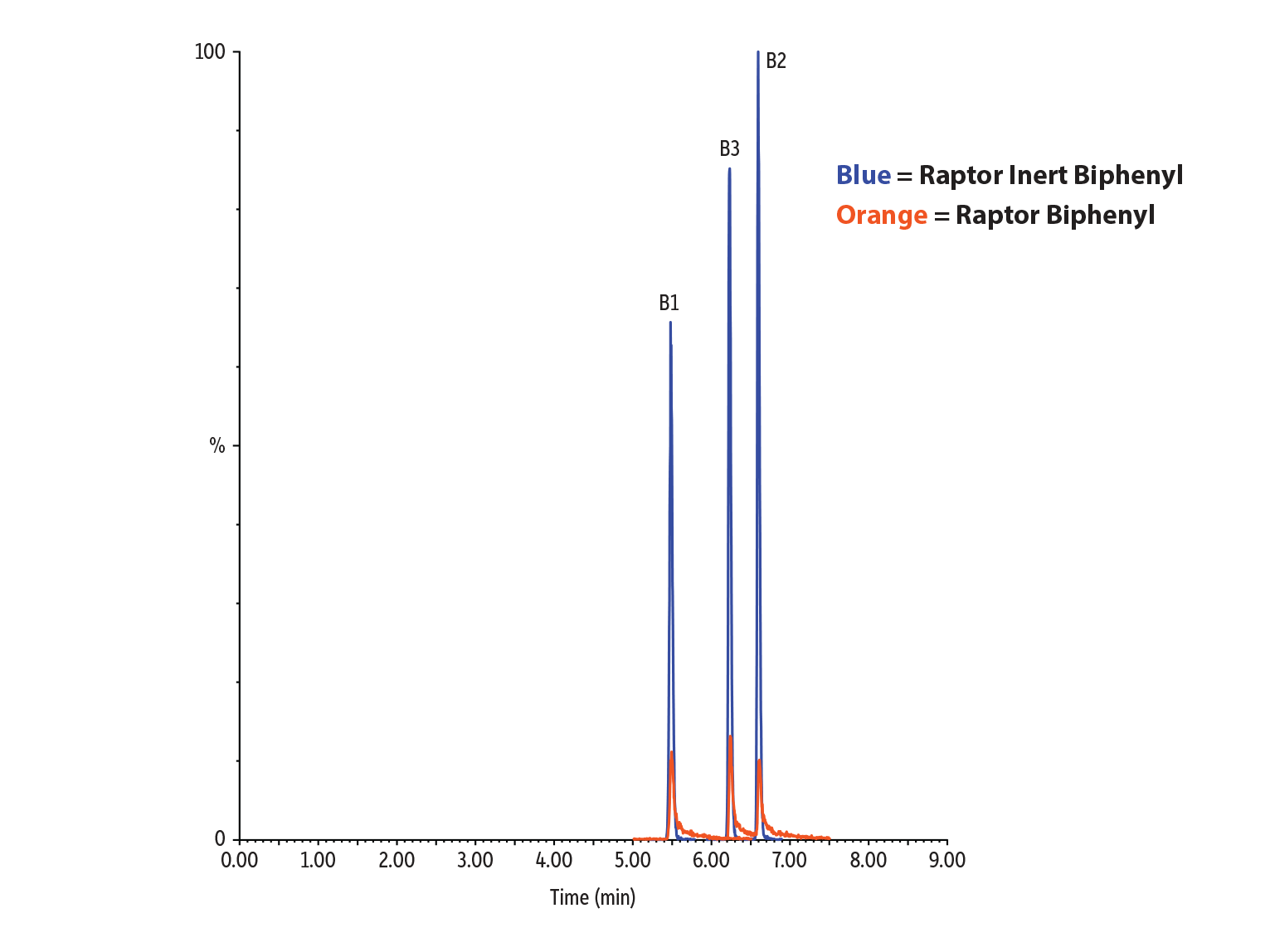
Figure 5: In this analysis of fumonisins, our new Raptor inert biphenyl columns achieve a dramatic increase in peak area compared to conventional, stainless-steel columns.
Figure 6: Peak area and peak height are greatly improved for the analysis of fumonisins.
Table II: In this analysis of mycotoxins, our new inert columns can provide an up to 10X increase in peak height over conventional columns.
| Compound | Peak Area | Peak Height | ||||
| Stainless Steel | Inert | Areas Ratio (Inert/Stainless Steel) | Stainless Steel | Inert | Height Ratio (Inert/Stainless Steel) | |
| Fumonisin B1 | 32578 | 122878 | 3.77 | 399544 | 2415567 | 6.05 |
| Fumonisin B2 | 23427 | 151719 | 6.48 | 383130 | 3869822 | 10.10 |
| Fumonisin B3 | 29864 | 143302 | 4.80 | 472279 | 3444421 | 7.29 |
| Ergocristine | 171197 | 195562 | 1.14 | 3865898 | 4450058 | 1.15 |
| Ergocristinine | 1393116 | 1583053 | 1.14 | 29212317 | 32975663 | 1.13 |
| Ergotamine | 433635 | 493003 | 1.14 | 8149518 | 9274156 | 1.14 |
| Ergotaminine | 397370 | 462119 | 1.16 | 7885403 | 9237991 | 1.17 |
| Ergocryptine | 446481 | 522204 | 1.17 | 9671753 | 11360839 | 1.17 |
| Ergocryptinine | 658788 | 778972 | 1.18 | 13680420 | 16765348 | 1.23 |
| Ergocornine | 370509 | 387025 | 1.04 | 7248981 | 7732744 | 1.07 |
| Ergocorninine | 590167 | 704029 | 1.19 | 12052359 | 14389283 | 1.19 |
| Ergosine | 445243 | 486620 | 1.09 | 8630932 | 9366602 | 1.09 |
| Ergosinine | 439026 | 496734 | 1.13 | 7820785 | 8740527 | 1.12 |
| T-2 | 43286 | 56535 | 1.31 | 1046233 | 1394735 | 1.33 |
| HT-2 | 10183 | 15221 | 1.49 | 216703 | 323765 | 1.49 |
| Tentoxin | 70973 | 95175 | 1.34 | 1577164 | 2131907 | 1.35 |
| Ochratoxin | 173686 | 190060 | 1.09 | 4039682 | 4411953 | 1.09 |
| Diacetoxyscirpenol | 47850 | 68139 | 1.42 | 846403 | 1208826 | 1.43 |
| Fusarenone X | 3865 | 7668 | 1.98 | 60409 | 121790 | 2.02 |
| 15-acetyl-DON | 17055 | 31369 | 1.84 | 269862 | 517570 | 1.92 |
| 3-acetyldeoxyvinalenol | 13353 | 22613 | 1.69 | 179204 | 296396 | 1.65 |
| Aflatoxin G2 | 171597 | 262824 | 1.53 | 3429501 | 5274354 | 1.54 |
| Aflatoxin G1 | 224058 | 304389 | 1.36 | 4607959 | 6102959 | 1.32 |
| ZON | 25617 | 37162 | 1.45 | 656915 | 927455 | 1.41 |
| Aflatoxin B2 | 159389 | 295648 | 1.85 | 3462489 | 5724754 | 1.65 |
| Aflatoxin B1 | 265935 | 223520 | 0.84 | 5335576 | 4425821 | 0.83 |
| Alpha-zearalenol | 16202 | 30224 | 1.87 | 382092 | 702420 | 1.84 |
| Deoxynivalenol | 6935 | 17346 | 2.50 | 117927 | 281906 | 2.39 |
| Nivalenol | 1790 | 4182 | 2.34 | 25276 | 64495 | 2.55 |
| Altenuene | 63224 | 113850 | 1.80 | 1187958 | 2059700 | 1.73 |
| Alternariol monomethyl ether | 19537 | 31024 | 1.59 | 428922 | 640689 | 1.49 |
| Alternariol | 48204 | 73272 | 1.52 | 837410 | 1302192 | 1.56 |
| Citrinin | 499900 | 1007880 | 2.02 | 5031182 | 9828890 | 1.95 |
| Tenuazonic acid | 21503 | 47828 | 2.22 | 89293 | 197658 | 2.21 |
Exceptional Inertness Meets Exceptional Stationary Phases
We’re introducing our inert LC column technology on three column types: Raptor Biphenyl, Raptor ARC-18, and Force Biphenyl. These new columns help bring the benefits of inert column technology to labs specializing in small molecule LC-MS/MS workflows.
Raptor LC Columns
Raptor LC columns combine the speed of 2.7 µm SPP with the resolution of Ultra Selective Liquid Chromatography (USLC) technology, improving separations and speeding up analysis times with standard HPLC instruments. When speed is your goal, Restek recommends the Raptor line of LC columns.
Learn more at www.restek.com/Raptor
Raptor Inert Biphenyl
The industry-leading Biphenyl is our most popular LC stationary phase. It is particularly adept at separating compounds that are hard to resolve or that elute early on C18 and other phenyl chemistries.
Raptor Inert ARC-18
The Raptor ARC-18 column features a well-balanced retention profile without the drawbacks of using an ordinary C18 in the harsh, acidic mobile phases needed for mass spectrometry. Even after extended use in these low-pH (≤ 2.0) conditions, the sterically protected ARC-18 offers consistent retention, peak shape, and response for charged bases, neutral acids, small polar compounds, and more.
Force LC Columns
Force fully porous particle (FPP) LC columns are designed and manufactured to handle high-pressure, high-stress conditions. They’re long-lasting, reproducible, and premium quality—backed by our 100% Pure Satisfaction guarantee. When you need greater retention and sharper peaks, Force LC columns are ready to be put to work.
Learn more at www.restek.com/Force
Force Inert Biphenyl
The Force Inert Biphenyl column separates compounds that other phenyl and C18 chemistries can’t. They allow the use of simple, MS-friendly mobile phases and are ideal for when you need to increase retention of hydrophilic aromatics.
Exceptional Inertness for Your Analysis
As chromatographers, we understand the importance of having confidence in your data. Whether you’re analyzing pesticides, mycotoxins, or any analysis containing metal-sensitive compounds, these new LC columns provide the accuracy, throughput, and performance your lab needs.
Contact your local Restek representative today at www.restek.com/contact-us to pair the benefits of our inert technology with your analysis.